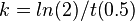Difference between revisions of "Degradation Pathways"
| (One intermediate revision by the same user not shown) | |||
| Line 1: | Line 1: | ||
[[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | [[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | ||
| − | + | To account for the decay in metabolite concentration over time, there are 46 decay reactions in this network. In these reactions, the product mass is pooled together under the term “Miscellaneous metabolites”. This is a collective term which refers to metabolites which are no longer of interest to this work, for example they are exported to the systemic circulation or undergo degradation. To account for the different rate of metabolite decay in the intracellular and extracellular compartments, each metabolite has two decay reactions which are governed by independent parameters. | |
| − | + | To calculate the rate constant, the following equation was used: | |
<math> k = ln(2)/t(0.5) </math> | <math> k = ln(2)/t(0.5) </math> | ||
Latest revision as of 09:51, 17 May 2019
To account for the decay in metabolite concentration over time, there are 46 decay reactions in this network. In these reactions, the product mass is pooled together under the term “Miscellaneous metabolites”. This is a collective term which refers to metabolites which are no longer of interest to this work, for example they are exported to the systemic circulation or undergo degradation. To account for the different rate of metabolite decay in the intracellular and extracellular compartments, each metabolite has two decay reactions which are governed by independent parameters.
To calculate the rate constant, the following equation was used: