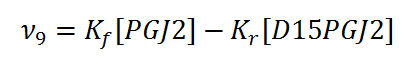Difference between revisions of "Transformation of PGJ2 to 15-D-PGJ2"
(→Parameters) |
|||
| Line 15: | Line 15: | ||
== Parameters == | == Parameters == | ||
| + | Note that the literature values are the same as [[Transformation of PGD2 to PGJ2 |reaction 8]]. | ||
| − | + | === Association Rate Constant (Kf) === | |
| + | |||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
| + | ! Value | ||
| + | ! Units | ||
| + | ! Conditions | ||
| + | ! Substrate | ||
| + | ! Reference | ||
| + | |- | ||
| + | |3.3E+6 (excluded) | ||
| + | |M-1 min-1 | ||
| + | |25°C and 0.055 ionic strength | ||
| + | |H2CO3 to CO2 | ||
| + | |<ref name="Gibbons1963”>[http://www.jbc.org/content/238/10/3502.full.pdf B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7]</ref> | ||
| + | |- | ||
| + | |4.5 | ||
| + | |M-1 min-1 | ||
| + | |20°C | ||
| + | |HC(OH)2COOH | ||
| + | |<ref name="Turyan1998”>[hrcak.srce.hr/file/195437 Y.I. Tur'yan, "Kinetics and Equilibrium of the Dehydration-Hydration and Recombination-Dissociation Reactions of Glyoxylic Acid Investigated by Electrochemical Methods", CCACAA 71 (3) 727¿743 (1998)]</ref> | ||
| + | |- | ||
| + | |0.42 | ||
| + | |M-1 min-1 | ||
| + | |20°C | ||
| + | |HC(OH)2COO– | ||
| + | |<ref name="Turyan1998”>[hrcak.srce.hr/file/195437 Y.I. Tur'yan, "Kinetics and Equilibrium of the Dehydration-Hydration and Recombination-Dissociation Reactions of Glyoxylic Acid Investigated by Electrochemical Methods", CCACAA 71 (3) 727¿743 (1998)]</ref> | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the reaction 9 Kf distribution | ||
| + | ! Mode (M-1 s-1) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 4.48E+00 || 1.09E+03 || 7.34E+00 || 2.42E+00 | ||
| + | |} | ||
| + | |||
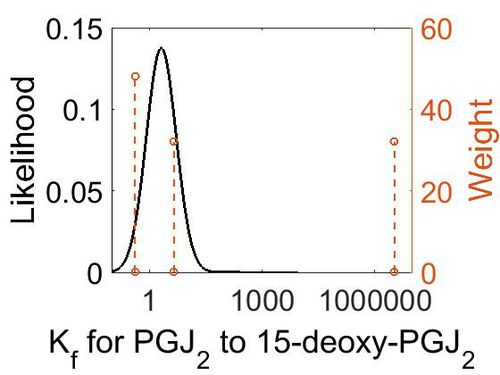
| + | [[Image:27.jpg|none|thumb|500px|The estimated probability distribution for reaction 9 Kf. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
| + | |||
| + | === Dissociation Rate Constant (Kr) === | ||
| + | This is a “Dependent parameter”, meaning that the log-normal distribution for this parameter was calculated using multivariate distributions (this is discussed in detail[[Quantification of parameter uncertainty | here]]). As a result, no confidence interval factor or literature values were cited for this parameter. | ||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the reaction 9 Kr distribution | ||
| + | ! Mode (M-1 s-1) !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 9.25E-04 || -6.07E+00 || 9.57E-01 | ||
| + | |- | ||
| + | |} | ||
| + | |||
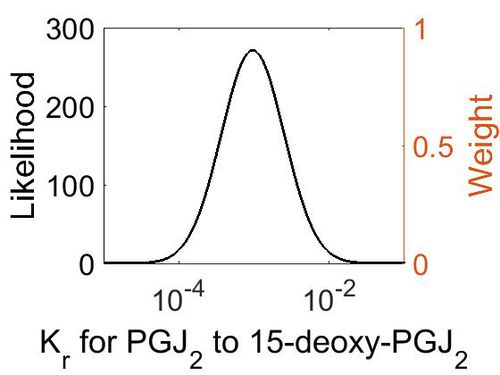
| + | [[Image:28.jpg|none|thumb|500px|The estimated probability distribution for reaction 9 Kr. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
| + | === Dissociation Constant === | ||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
| + | ! Value | ||
| + | ! Units | ||
| + | ! Conditions | ||
| + | ! Substrate | ||
| + | ! Reference | ||
| + | |- | ||
| + | |< 8E-04 | ||
| + | |N/A | ||
| + | |OH Radicals in the Presence of Added Gas Phase HNO3 (3 ppmv n-pentadecane, 0.25, 0.50, 1.0, or 2.0 ppmv HNO3, 10 ppmv O3, and 2 ppmv TME were added from a glass bulb in a flow of N2) | ||
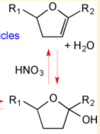
| + | |Acid-Catalyzed Dehydration of Cyclic Hemiacetals (n-Pentadecane) in SOA | ||
| + | [[File:Cyclic Hemiacetals.PNG |center|100px]] | ||
| + | |<ref name="Ranney2016”>[http://pubs.acs.org/doi/pdf/10.1021/acs.jpca.6b01402 A. Ranney "Kinetics of Acid-Catalyzed Dehydration of Cyclic Hemiacetals in Organic Aerosol Particles in Equilibrium with Nitric Acid Vapor" J. Phys. Chem. A, 2016, 120 (16), pp 2561–2568]</ref> | ||
| + | |- | ||
| + | |4E-04 | ||
| + | |N/A | ||
| + | |PH of 100 cc. 0-02N NaHCO3, saturated with C02, into which 0 95 cc. | ||
| + | |H2CO3 | ||
| + | |<ref name="BUYTENDYK1927”>[https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1251954/pdf/biochemj01144-0128.pdf F. BUYTENDYK "A Study of the System Carbonic Acid, Carbon Dioxide and Water - Determination of the True Dissociation-constant of Carbonic Acid" Biochem J. 1927; 21(3): 576–584.]</ref> | ||
| + | |- | ||
| + | |4.4E-4 | ||
| + | |N/A | ||
| + | |In a 0O008 mol. solution of carbonic acid at 4°, 1-23 % is present as H2CO3 | ||
| + | |H2CO3 | ||
| + | |<ref name="Thiel1914”>[http://onlinelibrary.wiley.com/doi/10.1002/cber.191404701173/epdf Thiel and Strohecker (1914). Ber. deutsch. chem. Gem. 47, 945, 1061.]</ref> | ||
| + | |- | ||
| + | |} | ||
| + | |||
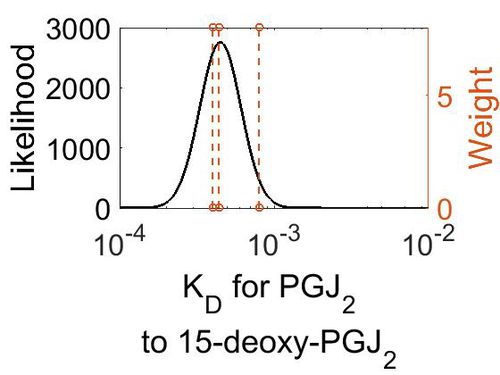
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the reaction 9 KD distribution | ||
| + | ! Mode (M-1 s-1) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 4.49E-04 || 1.38E+00 || -7.61E+00 || 3.07E-01 | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | [[Image:R9 kd.jpg|none|thumb|500px|The estimated probability distribution for reaction 9 KD. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
== Related Reactions == | == Related Reactions == | ||
* [[Transformation of PGD2 to PGJ2 |Transformation of PGD2 to PGJ2]] | * [[Transformation of PGD2 to PGJ2 |Transformation of PGD2 to PGJ2]] | ||
Revision as of 09:12, 15 May 2019
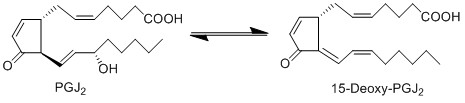
Due to the lack of PGD2 production by keratinocyte and fibroblast cells, the anti-inflammatory cyclopentanones, PGJ2 and 15d-PGJ2, will not be produced during early experimental procedures either.
Contents
Reaction
Chemical equation
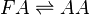
Rate equation
Parameters
Note that the literature values are the same as reaction 8.
Association Rate Constant (Kf)
| Value | Units | Conditions | Substrate | Reference |
|---|---|---|---|---|
| 3.3E+6 (excluded) | M-1 min-1 | 25°C and 0.055 ionic strength | H2CO3 to CO2 | [1] |
| 4.5 | M-1 min-1 | 20°C | HC(OH)2COOH | [2] |
| 0.42 | M-1 min-1 | 20°C | HC(OH)2COO– | [2] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 4.48E+00 | 1.09E+03 | 7.34E+00 | 2.42E+00 |
Dissociation Rate Constant (Kr)
This is a “Dependent parameter”, meaning that the log-normal distribution for this parameter was calculated using multivariate distributions (this is discussed in detail here). As a result, no confidence interval factor or literature values were cited for this parameter.
| Mode (M-1 s-1) | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|
| 9.25E-04 | -6.07E+00 | 9.57E-01 |
Dissociation Constant
| Value | Units | Conditions | Substrate | Reference |
|---|---|---|---|---|
| < 8E-04 | N/A | OH Radicals in the Presence of Added Gas Phase HNO3 (3 ppmv n-pentadecane, 0.25, 0.50, 1.0, or 2.0 ppmv HNO3, 10 ppmv O3, and 2 ppmv TME were added from a glass bulb in a flow of N2) | Acid-Catalyzed Dehydration of Cyclic Hemiacetals (n-Pentadecane) in SOA | [3] |
| 4E-04 | N/A | PH of 100 cc. 0-02N NaHCO3, saturated with C02, into which 0 95 cc. | H2CO3 | [4] |
| 4.4E-4 | N/A | In a 0O008 mol. solution of carbonic acid at 4°, 1-23 % is present as H2CO3 | H2CO3 | [5] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 4.49E-04 | 1.38E+00 | -7.61E+00 | 3.07E-01 |
Related Reactions
- Transformation of PGD2 to PGJ2
- ↑ B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7
- ↑ 2.0 2.1 [hrcak.srce.hr/file/195437 Y.I. Tur'yan, "Kinetics and Equilibrium of the Dehydration-Hydration and Recombination-Dissociation Reactions of Glyoxylic Acid Investigated by Electrochemical Methods", CCACAA 71 (3) 727¿743 (1998)]
- ↑ A. Ranney "Kinetics of Acid-Catalyzed Dehydration of Cyclic Hemiacetals in Organic Aerosol Particles in Equilibrium with Nitric Acid Vapor" J. Phys. Chem. A, 2016, 120 (16), pp 2561–2568
- ↑ F. BUYTENDYK "A Study of the System Carbonic Acid, Carbon Dioxide and Water - Determination of the True Dissociation-constant of Carbonic Acid" Biochem J. 1927; 21(3): 576–584.
- ↑ Thiel and Strohecker (1914). Ber. deutsch. chem. Gem. 47, 945, 1061.