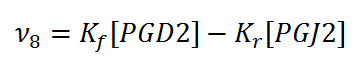Difference between revisions of "Transformation of PGD2 to PGJ2"
(→Parameters) |
(→Parameters) |
||
| Line 17: | Line 17: | ||
== Parameters == | == Parameters == | ||
| − | + | === Association Rate Constant (Kf) === | |
{|class="wikitable sortable" | {|class="wikitable sortable" | ||
| − | |+ style="text-align: left;" | | + | |+ style="text-align: left;" | Literature values |
|- | |- | ||
! Value | ! Value | ||
| Line 48: | Line 48: | ||
|} | |} | ||
| − | {|class="wikitable | + | {| class="wikitable" |
| − | |+ style="text-align: left;" | | + | |+ style="text-align: left;" | Description of the reaction 8 Kf distribution |
| + | ! Mode (M-1 s-1) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
|- | |- | ||
| − | + | | 4.48E+00 || 1.09E+03 || 7.34E+00 || 2.42E+00 | |
| − | + | |} | |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | | | + | === Dissociation Rate Constant (Kr) === |
| − | | | + | This is a “Dependent parameter”, meaning that the log-normal distribution for this parameter was calculated using multivariate distributions (this is discussed in detail[[Quantification of parameter uncertainty | here]]). As a result, no confidence interval factor or literature values were cited for this parameter. |
| − | | | + | {| class="wikitable" |
| − | | | + | |+ style="text-align: left;" | Description of the reaction 8 Kr distribution |
| − | + | ! Mode (M-1 s-1) !! Location parameter (µ) !! Scale parameter (σ) | |
|- | |- | ||
| + | | 9.25E-04 || -6.07E+00 || 9.57E-01 | ||
| + | |- | ||
|} | |} | ||
| + | === Dissociation Constant === | ||
{|class="wikitable sortable" | {|class="wikitable sortable" | ||
| − | |+ style="text-align: left;" | | + | |+ style="text-align: left;" | Literature values |
|- | |- | ||
! Value | ! Value | ||
| Line 93: | Line 97: | ||
|<ref name="Thiel1914”>[http://onlinelibrary.wiley.com/doi/10.1002/cber.191404701173/epdf Thiel and Strohecker (1914). Ber. deutsch. chem. Gem. 47, 945, 1061.]</ref> | |<ref name="Thiel1914”>[http://onlinelibrary.wiley.com/doi/10.1002/cber.191404701173/epdf Thiel and Strohecker (1914). Ber. deutsch. chem. Gem. 47, 945, 1061.]</ref> | ||
|- | |- | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the reaction 8 KD distribution | ||
| + | ! Mode (M-1 s-1) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 4.49E-04 || 1.38E+00 || -7.61E+00 || 3.07E-01 | ||
| + | |- | ||
|} | |} | ||
Revision as of 08:56, 15 May 2019
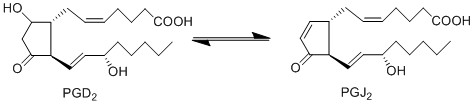
Due to the lack of PGD2 production by keratinocyte and fibroblast cells, the anti-inflammatory cyclopentanones, PGJ2 and 15d-PGJ2, will not be produced during early experimental procedures either.
Contents
Reaction
Chemical equation
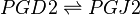
Rate equation
Parameters
Association Rate Constant (Kf)
| Value | Units | Conditions | Substrate | Reference |
|---|---|---|---|---|
| 3.3E+6 (excluded) | M-1 min-1 | 25°C and 0.055 ionic strength | H2CO3 to CO2 | [1] |
| 4.5 | M-1 min-1 | 20°C | HC(OH)2COOH | [2] |
| 0.42 | M-1 min-1 | 20°C | HC(OH)2COO– | [2] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 4.48E+00 | 1.09E+03 | 7.34E+00 | 2.42E+00 |
Dissociation Rate Constant (Kr)
This is a “Dependent parameter”, meaning that the log-normal distribution for this parameter was calculated using multivariate distributions (this is discussed in detail here). As a result, no confidence interval factor or literature values were cited for this parameter.
| Mode (M-1 s-1) | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|
| 9.25E-04 | -6.07E+00 | 9.57E-01 |
Dissociation Constant
| Value | Units | Conditions | Substrate | Reference |
|---|---|---|---|---|
| < 8E-04 | N/A | OH Radicals in the Presence of Added Gas Phase HNO3 (3 ppmv n-pentadecane, 0.25, 0.50, 1.0, or 2.0 ppmv HNO3, 10 ppmv O3, and 2 ppmv TME were added from a glass bulb in a flow of N2) | Acid-Catalyzed Dehydration of Cyclic Hemiacetals (n-Pentadecane) in SOA | [3] |
| 4E-04 | N/A | PH of 100 cc. 0-02N NaHCO3, saturated with C02, into which 0 95 cc. | H2CO3 | [4] |
| 4.4E-4 | N/A | In a 0O008 mol. solution of carbonic acid at 4°, 1-23 % is present as H2CO3 | H2CO3 | [5] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 4.49E-04 | 1.38E+00 | -7.61E+00 | 3.07E-01 |
Related Reactions
References
- ↑ B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7
- ↑ 2.0 2.1 [hrcak.srce.hr/file/195437 Y.I. Tur'yan, "Kinetics and Equilibrium of the Dehydration-Hydration and Recombination-Dissociation Reactions of Glyoxylic Acid Investigated by Electrochemical Methods", CCACAA 71 (3) 727¿743 (1998)]
- ↑ A. Ranney "Kinetics of Acid-Catalyzed Dehydration of Cyclic Hemiacetals in Organic Aerosol Particles in Equilibrium with Nitric Acid Vapor" J. Phys. Chem. A, 2016, 120 (16), pp 2561–2568
- ↑ F. BUYTENDYK "A Study of the System Carbonic Acid, Carbon Dioxide and Water - Determination of the True Dissociation-constant of Carbonic Acid" Biochem J. 1927; 21(3): 576–584.
- ↑ Thiel and Strohecker (1914). Ber. deutsch. chem. Gem. 47, 945, 1061.