ATP-Binding Cassette Transporters
The eicosanoids that are produced within the cell are immediately transported into the extracellular compartment, as they are cytotoxic \cite{Pompeia2002}. The transport systems relevant to each eicosanoid have not yet been fully characterised, therefore an ATP-binding cassette transporter has been assumed as the method of transportation across the cellular membrane for reactions 22-43. To model this we have used reversible the Michaelis Menten equation once more. The regeneration of ATP will be taken into account in the equilibrium constants. As there is no known data about the eicosanoid selectivity of the transporter, the Michaelis Menten constant (Km) and catalytic activity (kcat) are currently the same for each substrate.
Lipids are reported to be exported out of cells via an ATP binding cassette transporter. This process occurs against a concentration gradient; therefore hydrolysis of ATP is required to overcome the thermodynamic barrier.
Contents
Reaction
Chemical equation
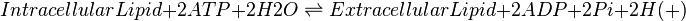
Rate equation
Parameters
| Value | Units | Species | Notes | Reference |
|---|---|---|---|---|
| 0.0109 ± 0.00391 | mM | Human | Substrate: LTC4
pH 7.0, 37°C, recombinant MRP2, using 4mM MgATP and 5g of isolated membranes. |
[1] |
| 0.0000366 ± 0.0000038 | mM | Human | Substrate LTC4, 37 °C, 4 mM ATP, | [2] |
| 5.3E-3 ± 2.6E-3 | mM | Human | 4 mM ATP, 37°C, 5–10 ml of membrane vesicle suspension (30
mg protein). |
[3] |
| Value | Units | Species | Notes | Reference |
|---|---|---|---|---|
| 11.34 | per minute | Human | Substrate: ATP | [1] |
| Value | Units | Species | Notes | Reference |
|---|---|---|---|---|
| 0.00019 ± 1.96e-5 | mmol/min/mg protein | Human | Substrate: LTC4
pH 7.0, 37°C, recombinant MRP2, using 4mM MgATP and 5g of isolated membranes. |
[1] |
| 1.25e-7 ± 1.2e-8 | mmol/min/mg | Human | Substrate LTC4, 37 °C, 4 mM ATP, | [2] |
| 2.02e-8 ± 5.9e-9 | mmol/mg/min | Human | 4 mM ATP, 37°C, 5–10 ml of membrane vesicle suspension (30
mg protein). |
[3] |
References
- ↑ 1.0 1.1 1.2 Yasunaga M. "Molecular cloning and functional characterization of cynomolgus monkey multidrug resistance-associated protein 2 (MRP2) Eur. J. Pharm. Sci. 35, 326-334 (2008) Cite error: Invalid
<ref>tag; name "Yas2007" defined multiple times with different content Cite error: Invalid<ref>tag; name "Yas2007" defined multiple times with different content - ↑ 2.0 2.1 Zeng H. "Transport of amphipathic anions by human multidrug resistance protein 3 J. Biol. Chem. 275, 34166-34172 (2000) Cite error: Invalid
<ref>tag; name "Mao2000" defined multiple times with different content - ↑ 3.0 3.1 "Functional reconstitution of substrate transport by purified multidrug resistance protein MRP1 (ABCC1) in phospholipid vesicles Cancer Res. 60, 4779-4784 (2000)