Difference between revisions of "Phosphofructokinase type 1"
| Line 12: | Line 12: | ||
\left (\left (\frac{\frac{[ATP]}{K_{ATP}}}{1 + \frac{[ATP]}{K_{ATP}} }\right ) \left ( \frac{ 1 + \frac{\beta Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } }{ 1 + \frac{Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } } \right ) | \left (\left (\frac{\frac{[ATP]}{K_{ATP}}}{1 + \frac{[ATP]}{K_{ATP}} }\right ) \left ( \frac{ 1 + \frac{\beta Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } }{ 1 + \frac{Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } } \right ) | ||
\left( \frac{\frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^3} | \left( \frac{\frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^3} | ||
| − | { \frac{L( 1 + \frac{CIT}{Ki_{CIT}})^4(1 + \frac{ATP}{Ki_{ATP}})^4}{(1+\frac{Fru2,6BP}{Ka_{Fru2,6BP}})^4} + \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^4 } \right)\right) | + | { \frac{L\left( 1 + \frac{CIT}{Ki_{CIT}}\right)^4\left(1 + \frac{ATP}{Ki_{ATP}}\right)^4}{\left(1+\frac{Fru2,6BP}{Ka_{Fru2,6BP}}\right)^4} + \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^4 } \right)\right) |
</math></center> | </math></center> | ||
Revision as of 17:36, 26 February 2014
The enzyme Phosphofructokinase Type-1 uses another ATP molecule to transfer a phosphate group to fructose 6-phosphate to form fructose 1, 6-bisphosphate. PFK-1 is an allosteric enzyme showing cooperative behaviour with Fru6P and hyperbolic kinetics with ATP.
Chemical equation
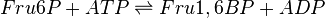
Rate equation
The concerted transition model of Monod, Wyman and Changeux (MWC model) is used as a rate equation for this tetrameric enzyme for considering exclusive ligand binding (F6P, activators and inhibitors) together with mixed type activation, (Fru2,6BP or AMP or Pi).
![\left (\left (\frac{\frac{[ATP]}{K_{ATP}}}{1 + \frac{[ATP]}{K_{ATP}} }\right ) \left ( \frac{ 1 + \frac{\beta Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } }{ 1 + \frac{Fru2,6BP}{ \alpha Ka_{Fru2,6BP} } } \right )
\left( \frac{\frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^3}
{ \frac{L\left( 1 + \frac{CIT}{Ki_{CIT}}\right)^4\left(1 + \frac{ATP}{Ki_{ATP}}\right)^4}{\left(1+\frac{Fru2,6BP}{Ka_{Fru2,6BP}}\right)^4} + \left[1 + \frac{Fru6P\left(1+\frac{Fru2,6BP}{\alpha Ka_{Fru2,6BP}}\right)}{K_{Fru6P\left(1 + \frac{Fru2n6BP}{Ka_{Fru2,6BP}}\right)}} \right]^4 } \right)\right)](/wiki/images/math/8/3/a/83a5f7224cd82998c6acfe1cc4fc1c77.png)