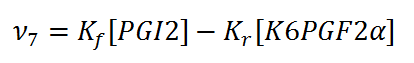Difference between revisions of "Transformation of PGI2 to 6-K-PGF1α"
(→Dissociation Constant) |
|||
| (One intermediate revision by the same user not shown) | |||
| Line 27: | Line 27: | ||
! Conditions | ! Conditions | ||
! Substrate | ! Substrate | ||
| + | ! Weight | ||
! Reference | ! Reference | ||
|- | |- | ||
| Line 34: | Line 35: | ||
Temperature: 25°C | Temperature: 25°C | ||
|3 | |3 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 41: | Line 43: | ||
Temperature: 25°C | Temperature: 25°C | ||
|3 | |3 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 48: | Line 51: | ||
Temperature: 25°C | Temperature: 25°C | ||
|4 | |4 | ||
| + | |48 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 55: | Line 59: | ||
Temperature: 25°C | Temperature: 25°C | ||
|4 | |4 | ||
| + | |48 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 62: | Line 67: | ||
Temperature: 25°C | Temperature: 25°C | ||
|5 | |5 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 69: | Line 75: | ||
Temperature: 25°C | Temperature: 25°C | ||
|6 | |6 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 76: | Line 83: | ||
Temperature: 25°C | Temperature: 25°C | ||
|7 | |7 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 83: | Line 91: | ||
Temperature: 25°C | Temperature: 25°C | ||
|8 | |8 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 90: | Line 99: | ||
Temperature: 25°C | Temperature: 25°C | ||
|8 | |8 | ||
| + | |32 | ||
|<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | |<ref name="Ross1982”>[http://pubs.acs.org/doi/pdf/10.1021/ja00370a035 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665]</ref> | ||
|- | |- | ||
| Line 96: | Line 106: | ||
| 25°C, in imidazole buffer and also in phosphate buffers, | | 25°C, in imidazole buffer and also in phosphate buffers, | ||
| CO2 to H2CO3 | | CO2 to H2CO3 | ||
| + | |32 | ||
| <ref name="Gibbons1963”>[http://www.jbc.org/content/238/10/3502.full.pdf B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7]</ref> | | <ref name="Gibbons1963”>[http://www.jbc.org/content/238/10/3502.full.pdf B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7]</ref> | ||
|- | |- | ||
| Line 129: | Line 140: | ||
! Conditions | ! Conditions | ||
! Substrate | ! Substrate | ||
| + | ! Weight | ||
! Reference | ! Reference | ||
|- | |- | ||
| Line 138: | Line 150: | ||
Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. | Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. | ||
|carbocyclic TXA2 (analogue of TXA2) | |carbocyclic TXA2 (analogue of TXA2) | ||
| + | |24 | ||
|<ref name="Alvarenga2010”>[http://journals.plos.org/plosbiology/article/file?id=10.1371/journal.pbio.1000547&type=printable P. H. Alvarenga "The Function and Three-Dimensional Structure of a Thromboxane A2/Cysteinyl Leukotriene-Binding Protein from the Saliva of a Mosquito Vector of the Malaria Parasite" PLoS Biol. 2010 Nov 30;8(11):e1000547. doi: 10.1371/journal.pbio.1000547.]</ref> | |<ref name="Alvarenga2010”>[http://journals.plos.org/plosbiology/article/file?id=10.1371/journal.pbio.1000547&type=printable P. H. Alvarenga "The Function and Three-Dimensional Structure of a Thromboxane A2/Cysteinyl Leukotriene-Binding Protein from the Saliva of a Mosquito Vector of the Malaria Parasite" PLoS Biol. 2010 Nov 30;8(11):e1000547. doi: 10.1371/journal.pbio.1000547.]</ref> | ||
|- | |- | ||
| Line 147: | Line 160: | ||
Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. | Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. | ||
|[3H]IONO NT-126, a TXA z antagonist, | |[3H]IONO NT-126, a TXA z antagonist, | ||
| + | |36 | ||
|<ref name="Nakahata1992”>[http://ac.els-cdn.com/S0006899310800137/1-s2.0-S0006899310800137-main.pdf?_tid=b2ab095e-155f-11e7-94f0-00000aab0f26&acdnat=1490888822_58d1d45dc3238dede94e4a2f15cada82 N Nakahata et al. "The Presence of Thromboxane A2 Receptors in Cultured Astrocytes From Rabbit Brain" Brain Res 583 (1-2), 100-104. 1992 Jun 26]</ref> | |<ref name="Nakahata1992”>[http://ac.els-cdn.com/S0006899310800137/1-s2.0-S0006899310800137-main.pdf?_tid=b2ab095e-155f-11e7-94f0-00000aab0f26&acdnat=1490888822_58d1d45dc3238dede94e4a2f15cada82 N Nakahata et al. "The Presence of Thromboxane A2 Receptors in Cultured Astrocytes From Rabbit Brain" Brain Res 583 (1-2), 100-104. 1992 Jun 26]</ref> | ||
|- | |- | ||
| Line 155: | Line 169: | ||
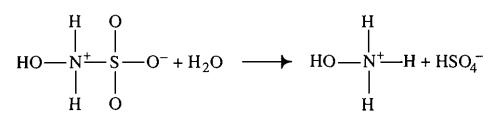
|Hydroxysulfamic acid | |Hydroxysulfamic acid | ||
[[File:HSA.PNG |center|500px]] | [[File:HSA.PNG |center|500px]] | ||
| + | |16 | ||
|<ref name="Littlejohn1989”>[http://www.nrcresearchpress.com/doi/pdf/10.1139/v89-243 D. LITTLEJOHN "The dissociation constant and acid hydrolysis rate of hydroxysulfamic acid" Can. J. Chem. 67, 1596 (1989).]</ref> | |<ref name="Littlejohn1989”>[http://www.nrcresearchpress.com/doi/pdf/10.1139/v89-243 D. LITTLEJOHN "The dissociation constant and acid hydrolysis rate of hydroxysulfamic acid" Can. J. Chem. 67, 1596 (1989).]</ref> | ||
|- | |- | ||
| Line 162: | Line 177: | ||
In vitro | In vitro | ||
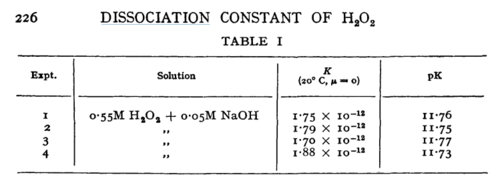
|H2O2 | |H2O2 | ||
| − | |<ref name=" | + | |16 |
| + | |<ref name="Evan1939”>[http://pubs.rsc.org/-/content/articlepdf/1949/tf/tf9494500224 M. G. EVAN "The dissociation constant and acid hydrolysis rate of hydroxysulfamic acid" Trans. Faraday Soc., 1949,45, 224-230]</ref> | ||
|- | |- | ||
|5.9 E-8 | |5.9 E-8 | ||
| Line 168: | Line 184: | ||
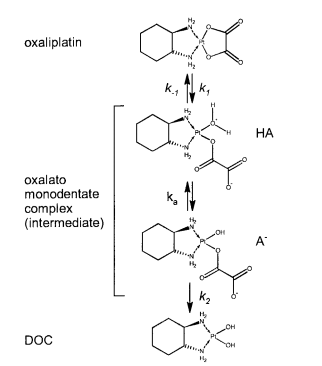
|Oxaliplatin | |Oxaliplatin | ||
|[[File:Oxaliplatin.PNG |center|500px]] | |[[File:Oxaliplatin.PNG |center|500px]] | ||
| + | |16 | ||
|<ref name="Jerremalm”>[http://www.sciencedirect.com/science/article/pii/S0022354916311649 Elin Jerremalm "Hydrolysis of Oxaliplatin—Evaluation of the Acid Dissociation Constant for the Oxalato Monodentate Complex" Journal of Pharmaceutical Sciences Volume 92, Issue 2, February 2003, Pages 436–438]</ref> | |<ref name="Jerremalm”>[http://www.sciencedirect.com/science/article/pii/S0022354916311649 Elin Jerremalm "Hydrolysis of Oxaliplatin—Evaluation of the Acid Dissociation Constant for the Oxalato Monodentate Complex" Journal of Pharmaceutical Sciences Volume 92, Issue 2, February 2003, Pages 436–438]</ref> | ||
|- | |- | ||
Latest revision as of 11:10, 2 November 2019
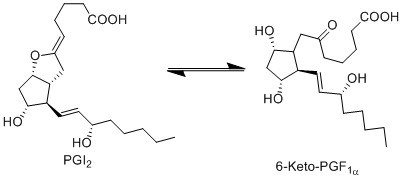
Due to ring strain, PGI2 is rapidly hydrolysed to 6-keto-PGF1a. This is a non-enzymatic reaction and results in the formation of a hydroxyl group at C9 and a ketone at C6, by incorporating two hydrogens and one oxygen.
Contents
Reaction
Chemical equation
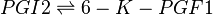
Rate equation
Parameters
Note that the literature values are the same as reaction 6.
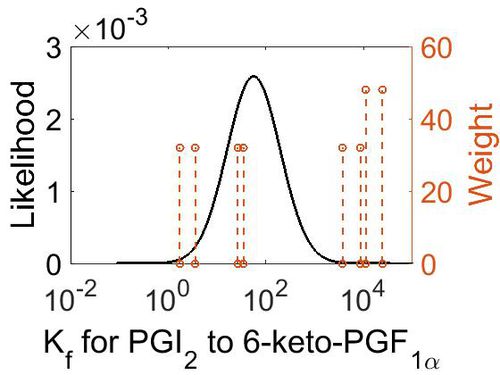
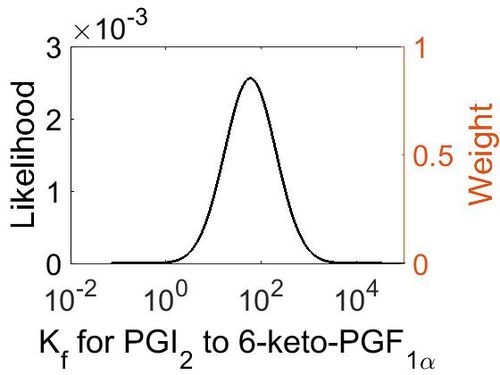
Association Rate Constant (Kf)
| Value | Units | Conditions | Substrate | Weight | Reference |
|---|---|---|---|---|---|
| 3.7e3 ± 0.1e3 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
3 | 32 | [1] |
| 8.7e3 | M-1 s-1 | KCl (1 M)
Temperature: 25°C |
3 | 32 | [1] |
| 1.1e4 ± 0.1e4 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
4 | 48 | [1] |
| 2.4 e4 | M-1 s-1 | KCl (1M)
Temperature: 25°C |
4 | 48 | [1] |
| 3.7e3 ± 0.1e3 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
5 | 32 | [1] |
| 3.6 ± 0.2 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
6 | 32 | [1] |
| 1.7 ± 0.1 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
7 | 32 | [1] |
| 26.7 ± 0.9 | M-1 s-1 | NaCl04 (0.1 - 0.2 M)
Temperature: 25°C |
8 | 32 | [1] |
| 35 | M-1 s-1 | KCl (1M)
Temperature: 25°C |
8 | 32 | [1] |
| 2.25 ± 0.12 | M-1 min-1 | 25°C, in imidazole buffer and also in phosphate buffers, | CO2 to H2CO3 | 32 | [2] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 3.70E+03 | 3.44E+01 | 1.10E+01 | 1.66E+00 |
Dissociation Rate Constant (Kr)
This is a “Dependent parameter”, meaning that the log-normal distribution for this parameter was calculated using multivariate distributions (this is discussed in detail here). As a result, no confidence interval factor or literature values were cited for this parameter.
| Mode | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|
| 5.28E+01 | 5.63E+00 | 1.29E+00 |
Dissociation Constant
| Value | Units | Conditions | Substrate | Weight | Reference |
|---|---|---|---|---|---|
| 0.000038 | mM | Temperature: 35°C
Vector:Mosquito Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. |
carbocyclic TXA2 (analogue of TXA2) | 24 | [3] |
| 0.00000023 | mM | Temperature: 35°C
Vector:Rabbit cultured astrocytes Note: "In solution, it undergoes rapid hydrolysis to form TXB2, a stable but physiologically inactive compound." - therefore they used stable analogues. |
[3H]IONO NT-126, a TXA z antagonist, | 36 | [4] |
| 1500 ± 500 (excluded) | mM | Temperature: 25°C
In vitro |
Hydroxysulfamic acid | 16 | [5] |
| N/A | Temperature: 20°C
In vitro |
H2O2 | 16 | [6] | |
| 5.9 E-8 | N/A | Oxaliplatin | 16 | [7] |
| Mode (M-1 s-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 2.35E-07 | 1.76E+04 | -6.86E+00 | 2.90E+00 |
Related Reactions
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 A. Ross "Vinyl epoxide hydrolysis reactions" J. Am. Chem. Soc., 1982, 104 (6), pp 1658–1665
- ↑ B. Gibbons "Rate of Hydration of Carbon Dioxide and Dehydration of Carbonic Acid at 25" J Biol Chem. 1963 Oct;238:3502-7
- ↑ P. H. Alvarenga "The Function and Three-Dimensional Structure of a Thromboxane A2/Cysteinyl Leukotriene-Binding Protein from the Saliva of a Mosquito Vector of the Malaria Parasite" PLoS Biol. 2010 Nov 30;8(11):e1000547. doi: 10.1371/journal.pbio.1000547.
- ↑ N Nakahata et al. "The Presence of Thromboxane A2 Receptors in Cultured Astrocytes From Rabbit Brain" Brain Res 583 (1-2), 100-104. 1992 Jun 26
- ↑ D. LITTLEJOHN "The dissociation constant and acid hydrolysis rate of hydroxysulfamic acid" Can. J. Chem. 67, 1596 (1989).
- ↑ M. G. EVAN "The dissociation constant and acid hydrolysis rate of hydroxysulfamic acid" Trans. Faraday Soc., 1949,45, 224-230
- ↑ Elin Jerremalm "Hydrolysis of Oxaliplatin—Evaluation of the Acid Dissociation Constant for the Oxalato Monodentate Complex" Journal of Pharmaceutical Sciences Volume 92, Issue 2, February 2003, Pages 436–438