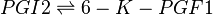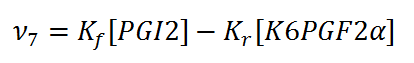Difference between revisions of "Transformation of PGI2 to 6-K-PGF1α"
| Line 1: | Line 1: | ||
[[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | [[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | ||
| + | |||
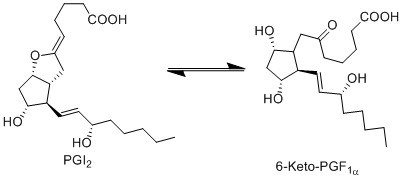
| + | Due to ring strain, PGI2 is rapidly hydrolysed to 6-keto-PGF1a. This is a non-enzymatic reaction and results in the formation of a hydroxyl group at C9 and a ketone at C6, by incorporating two hydrogens and one oxygen. | ||
| + | |||
== Reaction == | == Reaction == | ||
[[File:R7_PGI2_-_K6PGF2A.jpg |center|500px]] | [[File:R7_PGI2_-_K6PGF2A.jpg |center|500px]] | ||
Revision as of 17:16, 1 February 2019
Due to ring strain, PGI2 is rapidly hydrolysed to 6-keto-PGF1a. This is a non-enzymatic reaction and results in the formation of a hydroxyl group at C9 and a ketone at C6, by incorporating two hydrogens and one oxygen.
Reaction
Chemical equation