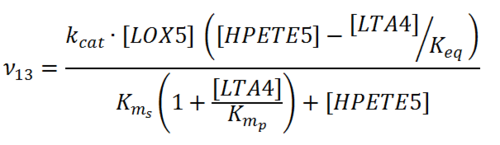Difference between revisions of "Transformation of 5-HPETE to LTA4"
(Created page with " Return to overview == Reaction == center|500px ==Chemical equation== <center...") |
|||
| (13 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
[[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | [[Welcome to the In-Silico Model of Cutaneous Lipids Wiki | Return to overview]] | ||
| + | |||
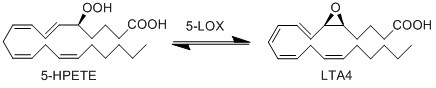
| + | In addition to being metabolised by PHGPx, 5-HPETE is also further metabolised by 5-LOX. This dehydration reaction converts the peroxide functional group of 5-HPETE to an epoxide functional group, generating LTA4 <ref>Shimizu, T. Radmark, O. Samuelsson, B., ''Enzyme with dual lipoxygenase activities catalyzes leukotriene A4 synthesis from arachidonic acid'', Proc Natl Acad Sci U S A (1984), 81, 689-93.</ref>. 5-LOX performs this reaction by abstracting the pro-R hydrogen at C10 and rearranging the structure so that the radical relocates to C6. The double bonds within the structure then rearrange to form a conjugated triene system and an epoxide. This reaction is promoted when 5-LOX colocalises with 5-lipoxygenase-activating protein (FLAP) on the nuclear membrane or the ER <ref>Abramovitz, M. Wong, E. Cox, M. E. Richardson, C. D. Li, C. Vickers, P. J. , ''5-lipoxygenase-activating protein stimulates the utilization of arachidonic acid by 5-lipoxygenase'', Eur J Biochem (1993), 215, 105-11.</ref><ref>Brock, T. G. Paine, R., 3rd Peters-Golden, M. , ''Localization of 5-lipoxygenase to the nucleus of unstimulated rat basophilic leukemia cells'', J Biol Chem (1994), 269, 22059-66.</ref>. | ||
| + | |||
| + | |||
== Reaction == | == Reaction == | ||
| Line 6: | Line 10: | ||
==Chemical equation== | ==Chemical equation== | ||
| − | <center><math> | + | <center><math> 5-HPETE \rightleftharpoons LTA4 </math></center> |
== Rate equation == | == Rate equation == | ||
| + | [[File:R13.PNG |center|500px]] | ||
== Parameters == | == Parameters == | ||
| + | Note that the literature values are the same as [[Transformation of AA to 5-HPETE|reaction 11]]. | ||
| + | |||
| + | === K<sub>ms</sub> === | ||
| + | |||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
| + | ! Value | ||
| + | ! Units | ||
| + | ! Species | ||
| + | ! Notes | ||
| + | ! Weight | ||
| + | ! Reference | ||
| + | |- | ||
| + | |5.10E-03 | ||
| + | |<math> mM </math> | ||
| + | |Human | ||
| + | |Expression Vector: Baculovirus, Sf9 insect cells | ||
| + | Enzyme: Recombinant 5-Lipoxygenase | ||
| + | pH: 5.6 | ||
| + | Temperature:37 | ||
| + | |256 | ||
| + | |<ref name="Shirumalla2006”>[http://www.ncbi.nlm.nih.gov/pubmed/17039282 Shirumalla R. K. “RBx 7,796: A novel inhibitor of 5-lipoxygenase.” Inflamm Res. 2006 Dec ; 55 (12) : 517-27.]</ref> | ||
| + | |- | ||
| + | |1.20E-02 | ||
| + | |<math> mM </math> | ||
| + | |Human | ||
| + | |Expression Vector: Polymorphonuclear Leukocytes | ||
| + | Enzyme: 5-Lipoxygenase | ||
| + | pH:7.5 | ||
| + | Temperature: 22 | ||
| + | |512 | ||
| + | |<ref name="Soberman1988"> [http://www.ncbi.nlm.nih.gov/pubmed/3070300 Soberman R. J. "5- and 15(omega-6)-lipoxygenases from human polymorphonuclear leukocytes.'' Methods Enzymol. 1988; 163:344-9.]</ref> | ||
| + | |- | ||
| + | |6.31E-02 | ||
| + | |<math> mM </math> | ||
| + | |Human | ||
| + | |Expression Vector: Polymorphonuclear Leukocytes | ||
| + | Enzyme: 5-Lipoxygenase | ||
| + | pH:7.5 | ||
| + | Temperature: 22 | ||
| + | |512 | ||
| + | |<ref name="Soberman1985”>[http://www.ncbi.nlm.nih.gov/pubmed/3920219 Soberman R. J. “Characterization and separation of the arachidonic acid 5-lipoxygenase and linoleic acid omega-6 lipoxygenase (arachidonic acid 15-lipoxygenase) of human polymorphonuclear leukocytes.” J Biol Chem. 1985 Apr 10;260(7):4508-15.]</ref> | ||
| + | |- | ||
| + | |} | ||
| + | |||
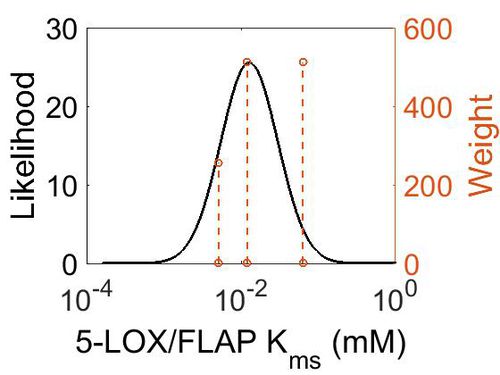
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the 5-LOX/FLAP Kms distribution | ||
| + | ! Mode (mM) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 1.27E-02 || 2.74E+00 || -3.76E+00 || 7.73E-01 | ||
| + | |} | ||
| + | |||
| + | [[Image:41.jpg|none|thumb|500px|The estimated probability distribution for 5-LOX/FLAP Kms. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
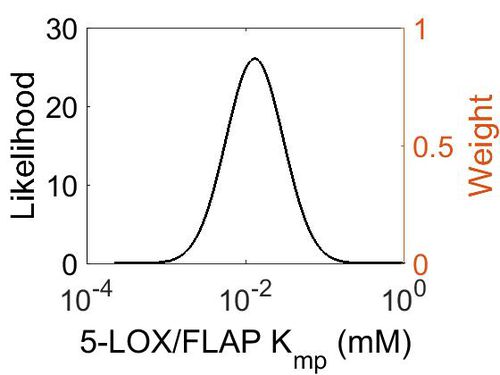
| + | === K<sub>mp</sub>=== | ||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the 5-LOX/FLAP Kmp distribution | ||
| + | ! Mode (mM) !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
| + | | 1.25E-02 || -3.63E+00 || 8.68E-01 | ||
| + | |} | ||
| + | |||
| + | [[Image:42.jpg|none|thumb|500px|The estimated probability distribution for 5-LOX/FLAP Kmp. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
| + | |||
| + | === k<sub>cat</sub> === | ||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
| + | ! Value | ||
| + | ! Units | ||
| + | ! Species | ||
| + | ! Notes | ||
| + | ! Weight | ||
| + | ! Reference | ||
| + | |- | ||
| + | |1500 + 75 | ||
| + | |per minute | ||
| + | |Potato | ||
| + | |Expression Vector:Potato Tubers | ||
| + | Enzyme: 5-Lipoxygenase | ||
| + | pH:5.5 | ||
| + | Temperature: 23 | ||
| + | |16 | ||
| + | |<ref name="Mulliez1987”>[http://www.sciencedirect.com/science/article/pii/0167483887902056 Mulliez E., “5-Lipoxygenase from potato tubers. Improved purification and physicochemical characteristics” Biochimica et Biophysica Acta, 1987;916(1):13-23.]</ref> | ||
| + | |- | ||
| + | |||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the 5-LOX/FLAP kcat distribution | ||
| + | ! Mode (min-1) !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
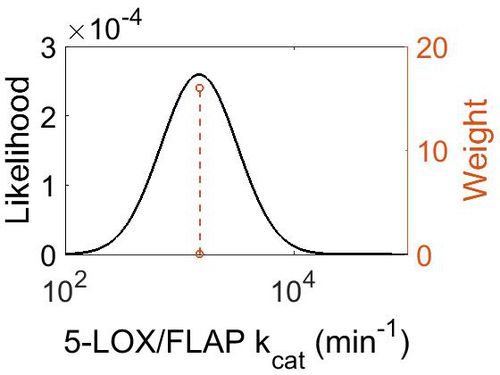
| + | | 1.50E+03 || 1.05E+00 || 7.31E+00 || 4.99E-02 | ||
| + | |} | ||
| − | {|class="wikitable" | + | [[Image:43.jpg|none|thumb|500px|The estimated probability distribution for 5-LOX/FLAP kcat. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] |
| − | + | ||
| + | |||
| + | === Enzyme concentration === | ||
| + | |||
| + | To convert the enzyme concentration from ppm to mM, the following [[Common equations#Enzyme concentration (mM)|equation]] was used. | ||
| + | |||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
! Value | ! Value | ||
! Units | ! Units | ||
! Species | ! Species | ||
| + | ! Notes | ||
| + | ! Weight | ||
| + | ! Reference | ||
| + | |- | ||
| + | |97.3 | ||
| + | |<math> ppm </math> | ||
| + | |Human | ||
| + | |Expression Vector: Lung | ||
| + | Enzyme: 5-LOX | ||
| + | pH: 7.5 | ||
| + | Temperature: 37 °C | ||
| + | |1024 | ||
| + | |<ref name="Kim2014"> [http://www.nature.com/nature/journal/v509/n7502/pdf/nature13302.pdf M. Kim ''A draft map of the human proteome'' Nature, 2014 509, 575–581]</ref> | ||
| + | |- | ||
| + | |49.8 | ||
| + | |<math> ppm </math> | ||
| + | |Human | ||
| + | |Expression Vector: Esophagus | ||
| + | Enzyme: 5-LOX | ||
| + | pH: 7.5 | ||
| + | Temperature: 37 °C | ||
| + | |1024 | ||
| + | |<ref name="Wilhelm2014"> [http://www.nature.com/nature/journal/v509/n7502/pdf/nature13319.pdf M. Wilhelm ''Mass-spectrometry-based draft of the human proteome'' Nature, 2014 509, 582–587]</ref> | ||
| + | |- | ||
| + | |31.9 | ||
| + | |<math> ppm </math> | ||
| + | |Human | ||
| + | |Expression Vector: Oral Cavity | ||
| + | Enzyme: 5-LOX | ||
| + | pH: 7.5 | ||
| + | Temperature: 37 °C | ||
| + | |1024 | ||
| + | |<ref name="Wilhelm2014"> [http://www.nature.com/nature/journal/v509/n7502/pdf/nature13319.pdf M. Wilhelm ''Mass-spectrometry-based draft of the human proteome'' Nature, 2014 509, 582–587]</ref> | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the 5-LOX concentration distribution | ||
| + | ! Mode (ppm) !! Mode (mM) !!Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
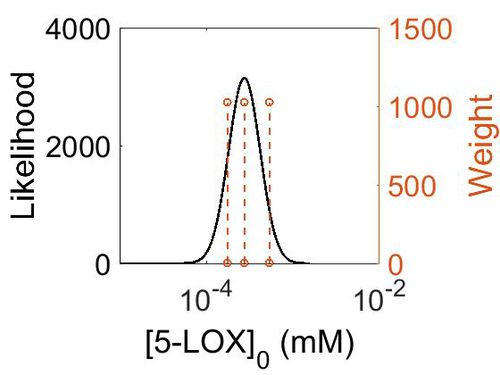
| + | | 4.96E+01 ||2.74E-04|| 1.60E+00 || 4.09E+00 || 4.28E-01 | ||
| + | |} | ||
| + | |||
| + | [[Image:158.jpg|none|thumb|500px|The estimated probability distribution for 5-LOX/FLAP concentration. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
| + | === K<sub>eq</sub> === | ||
| + | {|class="wikitable sortable" | ||
| + | |+ style="text-align: left;" | Literature values | ||
| + | |- | ||
| + | ! Gibbs Free Energy Change | ||
| + | ! Units | ||
| + | ! Species | ||
| + | ! Notes | ||
| + | ! Weight | ||
! Reference | ! Reference | ||
|- | |- | ||
| − | | | + | |(-86.007) |
| − | | | + | |kcal/mol |
| − | | | + | |Not stated |
| − | | | + | |Estimated |
| − | | | + | Enzyme: 5-LOX |
| + | Substrate: 5-HPETE | ||
| + | Product: LTA4 | ||
| + | pH: 7.3 | ||
| + | ionic strength: 0.25 | ||
| + | |64 | ||
| + | |<ref name="MetaCyc”>[http://metacyc.org/META/NEW-IMAGE?type=REACTION&object=RXN-13395 Caspi et al 2014, "The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of Pathway/Genome Databases," Nucleic Acids Research 42:D459-D471]</ref> | ||
|} | |} | ||
| + | {| class="wikitable" | ||
| + | |+ style="text-align: left;" | Description of the 5-LOX/FLAP Keq distribution | ||
| + | ! Mode !! Confidence Interval !! Location parameter (µ) !! Scale parameter (σ) | ||
| + | |- | ||
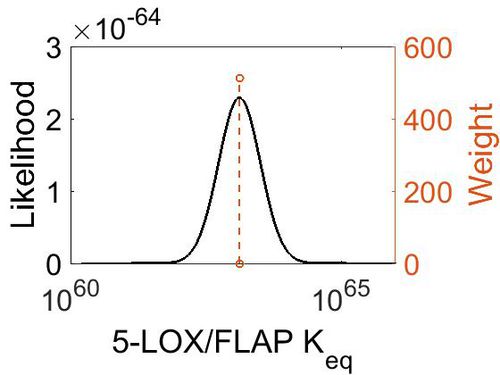
| + | | 1.31E+63 || 1.00E+01 || 1.46E+02 || 8.90E-01 | ||
| + | |} | ||
| + | |||
| + | [[Image:44.jpg|none|thumb|500px|The estimated probability distribution for 5-LOX/FLAP concentration. The value and weight of the literature values used to define the distribution are indicated by an orange dashed line. The x axis is plotted on a log-scale. ]] | ||
| + | |||
| + | == References == | ||
| + | <references/> | ||
== Related Reactions == | == Related Reactions == | ||
Latest revision as of 09:36, 21 August 2019
In addition to being metabolised by PHGPx, 5-HPETE is also further metabolised by 5-LOX. This dehydration reaction converts the peroxide functional group of 5-HPETE to an epoxide functional group, generating LTA4 [1]. 5-LOX performs this reaction by abstracting the pro-R hydrogen at C10 and rearranging the structure so that the radical relocates to C6. The double bonds within the structure then rearrange to form a conjugated triene system and an epoxide. This reaction is promoted when 5-LOX colocalises with 5-lipoxygenase-activating protein (FLAP) on the nuclear membrane or the ER [2][3].
Contents
Reaction
Chemical equation
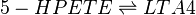
Rate equation
Parameters
Note that the literature values are the same as reaction 11.
Kms
| Value | Units | Species | Notes | Weight | Reference |
|---|---|---|---|---|---|
| 5.10E-03 | 
|
Human | Expression Vector: Baculovirus, Sf9 insect cells
Enzyme: Recombinant 5-Lipoxygenase pH: 5.6 Temperature:37 |
256 | [4] |
| 1.20E-02 | 
|
Human | Expression Vector: Polymorphonuclear Leukocytes
Enzyme: 5-Lipoxygenase pH:7.5 Temperature: 22 |
512 | [5] |
| 6.31E-02 | 
|
Human | Expression Vector: Polymorphonuclear Leukocytes
Enzyme: 5-Lipoxygenase pH:7.5 Temperature: 22 |
512 | [6] |
| Mode (mM) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 1.27E-02 | 2.74E+00 | -3.76E+00 | 7.73E-01 |
Kmp
| Mode (mM) | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|
| 1.25E-02 | -3.63E+00 | 8.68E-01 |
kcat
| Value | Units | Species | Notes | Weight | Reference |
|---|---|---|---|---|---|
| 1500 + 75 | per minute | Potato | Expression Vector:Potato Tubers
Enzyme: 5-Lipoxygenase pH:5.5 Temperature: 23 |
16 | [7] |
| Mode (min-1) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 1.50E+03 | 1.05E+00 | 7.31E+00 | 4.99E-02 |
Enzyme concentration
To convert the enzyme concentration from ppm to mM, the following equation was used.
| Value | Units | Species | Notes | Weight | Reference |
|---|---|---|---|---|---|
| 97.3 | 
|
Human | Expression Vector: Lung
Enzyme: 5-LOX pH: 7.5 Temperature: 37 °C |
1024 | [8] |
| 49.8 | 
|
Human | Expression Vector: Esophagus
Enzyme: 5-LOX pH: 7.5 Temperature: 37 °C |
1024 | [9] |
| 31.9 | 
|
Human | Expression Vector: Oral Cavity
Enzyme: 5-LOX pH: 7.5 Temperature: 37 °C |
1024 | [9] |
| Mode (ppm) | Mode (mM) | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|---|
| 4.96E+01 | 2.74E-04 | 1.60E+00 | 4.09E+00 | 4.28E-01 |
Keq
| Gibbs Free Energy Change | Units | Species | Notes | Weight | Reference |
|---|---|---|---|---|---|
| (-86.007) | kcal/mol | Not stated | Estimated
Enzyme: 5-LOX Substrate: 5-HPETE Product: LTA4 pH: 7.3 ionic strength: 0.25 |
64 | [10] |
| Mode | Confidence Interval | Location parameter (µ) | Scale parameter (σ) |
|---|---|---|---|
| 1.31E+63 | 1.00E+01 | 1.46E+02 | 8.90E-01 |
References
- ↑ Shimizu, T. Radmark, O. Samuelsson, B., Enzyme with dual lipoxygenase activities catalyzes leukotriene A4 synthesis from arachidonic acid, Proc Natl Acad Sci U S A (1984), 81, 689-93.
- ↑ Abramovitz, M. Wong, E. Cox, M. E. Richardson, C. D. Li, C. Vickers, P. J. , 5-lipoxygenase-activating protein stimulates the utilization of arachidonic acid by 5-lipoxygenase, Eur J Biochem (1993), 215, 105-11.
- ↑ Brock, T. G. Paine, R., 3rd Peters-Golden, M. , Localization of 5-lipoxygenase to the nucleus of unstimulated rat basophilic leukemia cells, J Biol Chem (1994), 269, 22059-66.
- ↑ Shirumalla R. K. “RBx 7,796: A novel inhibitor of 5-lipoxygenase.” Inflamm Res. 2006 Dec ; 55 (12) : 517-27.
- ↑ Soberman R. J. "5- and 15(omega-6)-lipoxygenases from human polymorphonuclear leukocytes. Methods Enzymol. 1988; 163:344-9.
- ↑ Soberman R. J. “Characterization and separation of the arachidonic acid 5-lipoxygenase and linoleic acid omega-6 lipoxygenase (arachidonic acid 15-lipoxygenase) of human polymorphonuclear leukocytes.” J Biol Chem. 1985 Apr 10;260(7):4508-15.
- ↑ Mulliez E., “5-Lipoxygenase from potato tubers. Improved purification and physicochemical characteristics” Biochimica et Biophysica Acta, 1987;916(1):13-23.
- ↑ M. Kim A draft map of the human proteome Nature, 2014 509, 575–581
- ↑ 9.0 9.1 M. Wilhelm Mass-spectrometry-based draft of the human proteome Nature, 2014 509, 582–587
- ↑ Caspi et al 2014, "The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of Pathway/Genome Databases," Nucleic Acids Research 42:D459-D471