Difference between revisions of "Serine out"
(→Reaction equation) |
(→Rate equation) |
||
| Line 7: | Line 7: | ||
==Rate equation== | ==Rate equation== | ||
| − | Simple mass action rate law is used. | + | Simple reversible mass action rate law is used. |
<center><math> v = K_{1} * [Serine] - K_{2} * [Serine_{out}] </math></center> | <center><math> v = K_{1} * [Serine] - K_{2} * [Serine_{out}] </math></center> | ||
Revision as of 09:20, 15 May 2014
This reaction describes the utilization of the endproduct Serine in other pathways.
Reaction equation
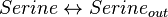
Rate equation
Simple reversible mass action rate law is used.
![v = K_{1} * [Serine] - K_{2} * [Serine_{out}]](/wiki/images/math/e/6/b/e6b7d292763cb679cbd1de648b4b0ddf.png)
Parameters
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|
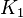
|
0.0103 [1] | 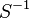
|
Escherichia coli |
References
- ↑ Turnaev II, Ibragimova SS, Usuda Y et al (2006). Mathematical modeling of serine and glycine synthesis regulation in Escherichia coli. Proceedings of the fifth international conference on bioinformatics of genome regulation and structure 2:78–83