Notes
Result generation
- Two types of simulation is being run. In one type of simulation, new parameter distribution is generated for each of the Control and silencing experiments. In the other type of simulation, first a parameter distribution is created and with that same parameter distribution the ensemble models of control and PYK(pyruvate kinase) silencing is being generated.
- When we silence the model using our skeleton model, the PEP(Phosphoenol pyruvate) accumulates and PYR(pyruvate) depletes. But when we calculate statistics from our ensemble of models generated with the parameter distribution, the average steady-state concentration shows a different behavior. In this mean value result, the PYK does deplete but there is no accumulation of PEP. Analyzing the results, we found that there is no specific parameter that is responsible for this undesired result rather it is due to different parameter distributions.
- When we silence the Pyruvate Kinase (dividing Vmax of PYK with 2 or 4 or 10) the PEP is supposed to accumulate and PYR is supposed to deplete. In our average value from the ensemble of models PYR does deplete but PEP does not accumulate. The rate laws has been verified and no mistake was found there. Therefore now the impact of other parameters on this silencing activity is being adjusted.
- We tried to first find out which factor is the most appropriate to divide the Vmax. We found that till factor 4 the PEP accumulates and after factor 4 although PEP concentration is higher than control but it is lower than the concentration we get by dividing with 4. So for silencing PYK we divided the Vmax with 4.
- We then generated 225 stead-states from control model using 300 total parameters. We used this 225 SS models to simulate steady-state for Pyruvate Kinase silenced by dividing Vmax with 4. This gives 202 SS models in PYK_Silence. We then deleted the No-steady state models of PYK_Silence from Control Data.
- We then wrote a Matlab script to filter the steady-state result from both Control and PYK_Silence for which the PEP concentration is higher in PYK_Silence compared to Control. We did this because we wanted to associate this fundamental information with our model and this is one of the criteria that the models should satisfy in order to perform correctly. This gives a total of 108 models where PEP is accumulated and Pyr is consumed in PYK_silence compared to Control
- We then took the parameter distribution associated with this 108 models to simulate the steady-state for different extracellular Serine concentration.
- For ease of notation we will designate the control model as shCntrl and the Pyruvate kinase silencing model as shPyk.
- Methods to calculate enzyme activity has been listed below in the table.
Model Checklist
- To find out the most likelihood factor for PYK silencing we designed an experiment to divide Vmax of PYK with numbers in between
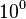 to
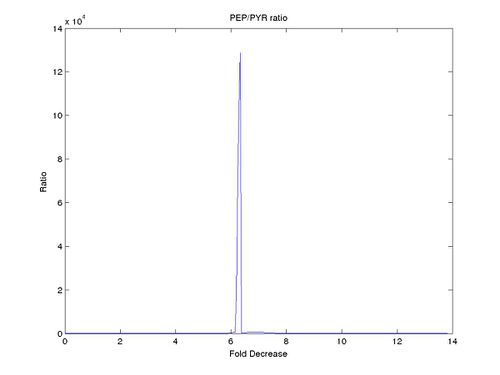
to 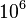 . Couple of interesting results were observed. The ratio of PEP/PYK increased until it reached a certain threshold after which there was a sharp decrease in the ratio as can be seen for one of the ensemble models (Model 23) in the figure below.
. Couple of interesting results were observed. The ratio of PEP/PYK increased until it reached a certain threshold after which there was a sharp decrease in the ratio as can be seen for one of the ensemble models (Model 23) in the figure below.
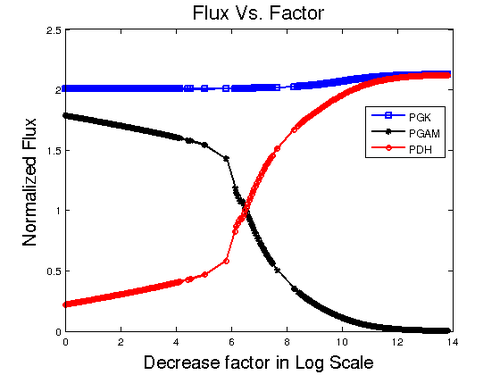
A close investigation with the result shows that silencing PYK causes 3PG to accumulate. In the pathway 3PG is entry point where the metabolite starts to accumulate. In the model three reactions are involved for production and consumption of 3PG. PGK (3-Phosphoglycerate kinase) is producing 3PG where as, PDH (Phosphoglycerate dehydrogenase) that goes in to Serine and Glycine biosynthesis and PGAM (3-phosphoglycerate mutase) which goes for Pyruvate and Lactate production are the two reactions that consume 3PG. To find out the reason for 3PG accumulation we plotted these three reaction against the division factor. The result is shown below.
As can be seen in the figure, the influx of PGK remains nearly constant. The flux to Serine biosynthesis grows higher with the increase of division factor and the flux to Pyruvate metabolism gets lower. Although 3PG is produced at the same rate, we can see that after 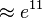 point, the PDH flux is almost the same as PGK meaning the production of 3PG is same as the consumption which explains why the concentration of 3PG goes down after a sudden increase. To explain the accumulation, the production flux for 3PG remains constant, but the consumption flux (PDH + PGAM) is still low compared with production. After the crossing point at
point, the PDH flux is almost the same as PGK meaning the production of 3PG is same as the consumption which explains why the concentration of 3PG goes down after a sudden increase. To explain the accumulation, the production flux for 3PG remains constant, but the consumption flux (PDH + PGAM) is still low compared with production. After the crossing point at 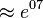 the PDH starts to increase meaning that the consumption of 3PG increases which levels with PGK at
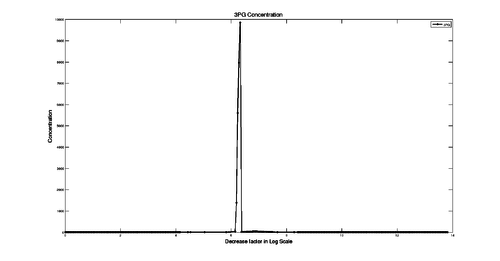
the PDH starts to increase meaning that the consumption of 3PG increases which levels with PGK at 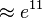 . The figure below displays the 3PG concentration over factor decrease.
. The figure below displays the 3PG concentration over factor decrease.
A checklist for model validation is given below
| Criteria | Result | Comments |
|---|---|---|
| The concentration of PEP should be higher than Pyruvate in Control Model. | YES | |
| In the warburg effect, energy production is shifted from pyruvate metabolism to lactate production. To imitate this physiological phenomena in our model, the Lactate dehydrogenase (LDH) should have higher flux compared to Mitochondrial pyruvate metabolism (MPM). In literature it is reported that the value is ~11.8 times higher. | YES | |
| Acorrding to Chaneton et. al. [1] when the pyruvate kinase is silenced, the phosphoenol pyruvate (PEP) should be accumulating. | YES (with observation) | The average value calculated from 108 ensemble models shows the PEP concentration in shCntrl as 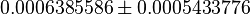 and in shPyk as and in shPyk as 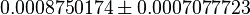 indicating PEP accumulation in the shPyk. However, the paper reported a 100 fold increase in PEP in their experiment whereas in our model it is only ~1.4 fold. indicating PEP accumulation in the shPyk. However, the paper reported a 100 fold increase in PEP in their experiment whereas in our model it is only ~1.4 fold.
|
| Acorrding to Chaneton et. al. [1] ~50% decrease is reported in Pyruvate levels in shPyk. | YES | In our model, the concentration of Pyruvate in shCntrl is 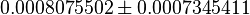 and in shPyk the concentration is and in shPyk the concentration is 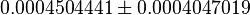 showing a decrease in Pyruvate for shPyk. In our model the decrease is ~44%. showing a decrease in Pyruvate for shPyk. In our model the decrease is ~44%.
|
| Chaneton et. al. [1] also reported that Blocking PYK activity shifted the metabolism of glucose away from lactate production to citrate production in the mitochondria. This is shown in Figure 2 of the paper where cumulative intensities of each of the metabolite isotopomers was analyzed at the indicate time point. In the figure it can be seen that the cummulative intensity of Lactate is lower in shPyk compared to shCntrl. | YES | In our model, the intracellular steady-state Lactate concentration of shCntrl is 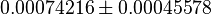 and for shPyk it is and for shPyk it is 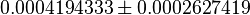 . This shows that the lactate production is decreased in shPyk compared to shCntrl as shown in the paper. Moreover, the MPM/LDH flux ratio gives a value of 0.0002846206 for shCntrl and 0.0081609042, showing that the flux through MPM is higher in shPyk compared with shCntrl. . This shows that the lactate production is decreased in shPyk compared to shCntrl as shown in the paper. Moreover, the MPM/LDH flux ratio gives a value of 0.0002846206 for shCntrl and 0.0081609042, showing that the flux through MPM is higher in shPyk compared with shCntrl.
|
| Iqbal (2012) et. al.[2] reported a decreased Glucose uptake on shPyk. | YES | In our model, we also observe a decrease in Glucose uptake for shPyk. The Glucose uptake (GLUT) flux for shPyk is 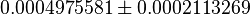 and for shCntrl it is and for shCntrl it is 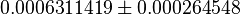 showing a lower value for GLUT in shPyk. showing a lower value for GLUT in shPyk.
|
| Chaneton et. al. [1] reported an increased metabolic flux into the serine and glycine biosynthesis pathway with reduced pyruvate kinase activity. | No/YES (with observation) | The steady state concentration of Serine and Glycine is low in shPyk than in shCntrl. The intracellular Serine and Glycine concentration in shPyk is Failed to parse (Cannot store math image on filesystem.): 7.04901E-005 \pm 3.318E-005 and Failed to parse (Cannot store math image on filesystem.): 0.0001577779 \pm 0.0001046952 whereas in ShCntrl it is Failed to parse (Cannot store math image on filesystem.): 7.7776E-005 \pm 3.596E-005 and Failed to parse (Cannot store math image on filesystem.): 0.0001729984 \pm 0.000115717 respectively. The Serine and Glycine concentration contradicts with the findings of Chaneton et. al. [1] if we consider only the steady-state concentration. However, in our model the Glucose uptake flux is lower in shPkm compared to shCntrl. Normalizing the flux with respect to Glucose uptake for shCntrl and shPyk shows a higher flux through Phosphoglycerate dehydrogenase (PDH) the entry point for the Serine and Glycine. The normalized flux value of PDH for shCntrl is 1.5561722231 and for shPyk it is 1.7719215457 indicating a higher flux in to the production of Serine and Glycine with respect to Glucose uptake in shPyk. |
| Chaneton et. al. [1] reported that the absence of extracellular serine and glycine had a pronounced inhibitory effect on pyruvate kinase activity. | YES | To verify this statement we designed an experiment where we made the SerineOUT = 0 in our model. The normalized flux value for shSerine_0 shows that PYK has a flux value of 0.6101033422 whereas for Control the flux is 0.6104158651 indicating a slight inhibition in the absence of extracelluar Serine. |
| Chaneton et. al. [1] also reported that when serine and glycine starved cell were incuvated with serine, intracellular pyruvate kinase activity was increased relative to starved cells. | YES | To verify this statement we designed an experiment where we made the SerineOUT = 50 in our model. The normalized flux value for shSerine_50 shows that PYK has a flux value of 2.0327570679 whereas for shSerine_0 the flux is 0.6101033422 indicating an increase in PYK activity in the presence of extracelluar Serine. |
| Glycine did not simulate intracellular pyruvate kinase activity. | YES | To verify this statement we designed an experiment where we made the GlycineOUT = 50 where keeping the value of SerineOUT = 0 in our model. The normalized flux value for shGlycine_50 shows that PYK has a flux value of 0.6225994227 whereas for Control the flux is 0.6104158651 indicating very little effect in Pyruvate kinase in the presence of high Glycine. |
| Serine was demonstrated to activate recombinant PYK with a half-maximal activation concentration of 1.3 mM. | ||
| Serine concentration in the blood is ~20 fold below that of Glucose | ||
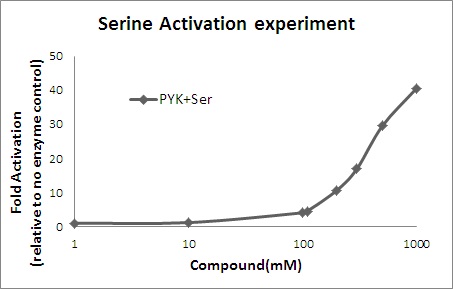
| Fold activation of pyruvate kinase | YES | To verfity the fold activation with respect to no enzyme control we designed an experiment where we ran steady-state with Serince concentration, 0.001, 0.01, 0.1, 1 ,10, 100, 1000 mM. Number of steady-states reached with those values are 102, 89, 46, 77, 99, 97, 92. The following two figures show the comparative result. The data was collected at the time point of 12 Sec. Later extra data was generated for 110, 300 and 500 mM of Serine |
| Glycogen synthesis and degradation is lower compared to Glycolytic flux (LDH) in cancer cells. | YES (With Observation) | Hernandez et. al. (2011)[3] repored that the Glycogen synthesis and degradation is 10-18 times lower in cancer cells. In our model it is lower at much higher fold (~1800). |
Observation
The following observations are made from the model
| Observation | Comments |
|---|---|
| What is the observation on ATP production? Is it higher in shCntrl or shPYK or in Serine_10000? | |
| Does excessive extracellular Serine redirect energy production from Lactate production to Mitochondrial pyruvate metabolism? Does this solves the Warburg effect? | Increase in extracellular Serine production does shift energy metabolism from Lactate production to Mitochondrial pyruvate metabolism. The ratio with normalized fluxes of MPM/LDH in control was 0.0002846 whereas with 50 mM extracellular Serine it becomes 0.651727 clearly indicating a large shift from LDH to MPM. However, still the flux through LDH is higher than MPM although now with a small factor (1.53 ) compared to very high factor in Control (3513.44). Important Observation: when the extracellular Serine concentration is 1 then the flux totally shifts from LDH to MPM as the ratio becomes (0.7741). Point to note: Two things might cause this shift in flux. Point 1: This could be an artifact of the model itself. When extracellular concentration of Serine is 1 mM only 76 out of 108 steady states were found whereas for all other experiments no. of steady-state was above 90. Point 2: It could still indicate that there might be a point of threshold in Serine concentration that can be used to completely shift the glycolytic metabolism from Lactate production to mitochondrial pyruvate metabolism as reported by Chaneton et. al. [1], that Blocking PYK activity shifted the metabolism of glucose away from lactate production to citrate production in the mitochondria. |
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Barbara Chaneton, Petra Hillmann, Liang Zheng, Agnès C. L. Martin, Oliver D. K. Maddocks, Achuthanunni Chokkathukalam, Joseph E. Coyle, Andris Jankevics, Finn P. Holding, Karen H. Vousden, Christian Frezza, Marc O’Reilly & Eyal Gottlieb (2012). Serine is a natural ligand and allosteric activator of pyruvate kinase M2, Nature, 491:458–462 (doi)
- ↑ Iqbal MA, Bamezai RNK (2012), Resveratrol Inhibits Cancer Cell Metabolism by Down Regulating Pyruvate Kinase M2 via Inhibition of Mammalian Target of Rapamycin, PLoS ONE 7(5): e36764. (doi)
- ↑ Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011). Modeling cancer glycolysis. Biochim Biophys Acta, 1807:755–767 (doi)