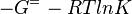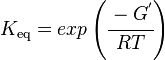Limonene Synthase
You can go back to main page of the kinetic model here.
Contents
What we know
Issues
Strategies
Reaction catalysed
- geranyl diphosphate Failed to parse (Cannot store math image on filesystem.): \rightleftharpoons (−)-(4S)-limonene + diphosphate
Metabolite Background Information
Long metabolite names are abbreviated in the model for clarity and standard identification purposes.
| Metabolite | Abbreviation | Chemical Formula | Molar mass (g/mol) | ChEBI | ChEMBL | PubChem |
|---|---|---|---|---|---|---|
| Geranyldiphosphate | GPP | C10H20O7P2 | 314.209 | 17211 | 41432 | 445995 |
| Limonene | Limonene | C10H16 | 136.24 | 15384 | 449062 | 22311 or 439250 |
| Diphosphate | PP | O7P2 | 173.94 | 644102 |
Equation Rate
- Failed to parse (Cannot store math image on filesystem.): V_\mathrm{LimSynth} = Vmax_\mathrm{forward} * \cfrac {\cfrac{[GPP]}{Km_\mathrm{GPP}} * \left ( 1 - \cfrac {[Limonene]*[PP]}{[GPP]*K_\mathrm{eq}} \right )}{\left (1 + \cfrac {[GPP]}{Km_\mathrm{GPP}} + \cfrac {[Limonene]}{Km_\mathrm{Limonene}} \right) * \left (1 + \cfrac {[PP]}{Km_\mathrm{PP}} \right)}
| Parameter | Description | Reference |
|---|---|---|
| VLimSynth | Reaction rate for Limonene Synthase | ref |
| Vmaxforward | Maximum reaction rate towards the production of limonene | ref |
| KmGPP | Michaelis-Menten constant for GPP | ref |
| KmLimonene | Michaelis-Menten constant for Limonene | ref |
| KmPP | Michaelis-Menten constant for PP | ref |
| Keq | Equilibrium constant | ref |
| [GPP] | GPP concentration | ref |
| [Limonene] | Limonene concentration | ref |
| [PP] | PP concentration | ref |
Strategies for estimating the kinetic parameter values
Calculating the Equilibrium Constant
The equlibrium constant, Keq, can be calculated from the Gibbs free energy of a reaction, ΔGr, using the Van't Hoff isotherm equation:
and by dividing both sides of the equation with RT, and later take the exponents of both sides, the Keq can be calculated by this equation:
where;
| Keq | Equilibrium constant |
| -ΔG° | Gibbs free energy change |
| R | Gas constant with a value of 8.31 JK-1mol-1 |
| T | Temperature which is always expressed in kelvin |
Extracting Information from Limonene Production Rates
| Amount produced (mg/L) | Time (H) | Organism | Description | Reaction Flux (µM/s) |
|---|---|---|---|---|
| 5 | 24 | Escherichia coli | Possible reason for the low limonene production might due to the insufficient supply of IPP and DMAPP [1]. | 0.0255 |
| 335 | 48 | Escherichia coli | Engineered E.coli in which heterologous MVA pathway was installed [2]. | 0.8537 |
| 35.8 | 48 | Escherichia coli | E.coli was engineered to express GPPS, LS, DXS, and IDI [3] . | 0.0912 |
| 4.87 | 48 | Escherichia coli | This was the initial titer. The study established a limonene biosynthesis pathway in E.coli using four different polycistronic operons based on 3 vectors with varied expression strength [4]. | 0.0124 |
| 17.4 | 48 | Escherichia coli | Using a plasmid with DXS and IDI over expressed [5]. | 0.0445 |
| 430 | 72 | Escherichia coli | [6] | 0.7306 |
Published Kinetic Parameter Values
| Km (mM) | Vmax | Kcat (s-1) | Kcat/Km | Organism | Description |
|---|---|---|---|---|---|
| 0.00125 | - | - | - | Ricciocarpos natans | GPP -> Limonene |
| 0.0018 | - | - | - | Mentha piperita | GPP -> Limonene |
| 0.00625 | 0.08 µmol/min/mg | 0.08 | 1.5 | Cannabis sativa L. | GPP -> Limonene |
| 0.00496 | 0.13 µmol/min/mg | 0.14 | 2.9 | Cannabis sativa L. | GPP -> Limonene |
| 0.0031 | 28.49 µmol/h/mg | - | - | Citrus limon | GPP -> Limonene |
| 0.016 | - | 0.02 | - | Escherichia coli | GPP -> Limonene |
| 0.0068 | - | 0.082 | - | Cannabis sativa L. | GPP -> Limonene |
| 0.0067 | - | 0.081 | - | Cannabis sativa L. | GPP -> Limonene |
Simulations
References
- ↑ Carter, Ora A. et. al.2013. "Monoterpene biosynthesis pathway construction in Escherichia coli",Phytochemistry, 64:425–433, 2003.
- ↑ Alonso-Gutierez et. al. 2013. "Metabolic engineering of Escherichia coli for limonene and perillyl alcohol production", Metabolic Engineering, 19:33-41
- ↑ Du et. al. 2014. "Enhanced limonene production by optimizing the expression of limonene biosynthesis and MEP pathway genes in E.coli", Bioprocessing and Bioprocessing, 1:10
- ↑ Du et. al. 2014. "Enhanced limonene production by optimizing the expression of limonene biosynthesis and MEP pathway genes in E.coli", Bioprocessing and Bioprocessing, 1:10
- ↑ Du et. al. 2014. "Enhanced limonene production by optimizing the expression of limonene biosynthesis and MEP pathway genes in E.coli", Bioprocessing and Bioprocessing, 1:10
- ↑ Alonso-Gutierez et. al. 2013. "Metabolic engineering of Escherichia coli for limonene and perillyl alcohol production", Metabolic Engineering, 19:33-41