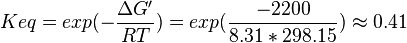Hexose-6-phosphate isomerase
The enzyme Hexose-6-phosphate isomerase converts Glucose 6-phosphate (Glc6P) into its isomer Fructose 6-phosphate (Fru6P). Isomers have the same molecular formula, but the atoms of each molecule are arranged differently.
Contents
Chemical reaction
Rate equation
Reversible competitive inhibition with Ery4P, 6PG and FBP. [1]
![v=\frac{V_{mf}\frac{[Glc6P]}{Km_{Glc6P}} - V_{mr} \frac{[Fru6P]}{Km_{Fru6P}} }{ 1 + \frac{[Glc6P]}{Km_{Glc6P}} + \frac{[Fru6P]}{Km_{Fru6P}} + \frac{[ERY4P]}{Ki_{ERY4P}} + \frac{[6PG]}{Ki_{6PG}} + \frac{[Fru1,6BP]}{Ki_{Fru1,6BP}} }](/wiki/images/math/4/f/d/4fd18a682ee7d511c57c5db14dc09b43.png)
Substituting with Haldane equation to ensure thermodynamic consistency we have
![v= \frac{V_{mf}\frac{[Glc6P]}{Km_{Glc6P}}\left(1 - \frac{[Fru6P]}{K_{eq}[Glc6P]} \right)}{ 1 + \frac{[Glc6P]}{Km_{Glc6P}} + \frac{[Fru6P]}{Km_{Fru6P}} + \frac{[ERY4P]}{Ki_{ERY4P}} + \frac{[6PG]}{Ki_{6PG}} + \frac{[Fru1,6BP]}{Ki_{Fru1,6BP}} }](/wiki/images/math/d/0/d/d0dd6e9a5e0114020d8f8122d107db36.png)
Parameter values
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.4 [1] | 
|
HeLa cell line | 3 cell assays |
|
0.9[1] | 
|
1 cell assay | |
|
0.4 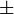 0.03 mM[1] 0.03 mM[1]
|
mM | 3 cell assays | |
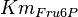
|
0.05[1] | mM | 2 cell assays | |
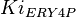
|
0.001[1] | mM | Adjusted in the interval based on activity | |
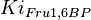
|
0.06[1] | mM | Adjusted in the interval based on activity | |
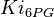
|
0.015[1] | mM | Adjusted in the interval based on activity |
Parameters with uncertainty
- The V is measured in the reverse order for the enzyme (
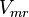 ) in [2] as
) in [2] as 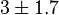 . The
. The  is calculated based on Haldane equation. The same relative percent error for
is calculated based on Haldane equation. The same relative percent error for 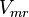 is used to define the uncertainty of this parameter. The value is
is used to define the uncertainty of this parameter. The value is 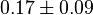
- The uncertainty of
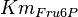 for HeLa cell line are calculated based on the same proportion of uncertainty for AS-30D cell reported in the paper Marín-Hernández et. al., Modeling cancer glycolysis [1].
for HeLa cell line are calculated based on the same proportion of uncertainty for AS-30D cell reported in the paper Marín-Hernández et. al., Modeling cancer glycolysis [1].
- The value of
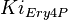 ,
, 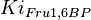 and
and 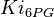 are the averaged from the forward and reverse reaction value listed in Table S6 [1].
are the averaged from the forward and reverse reaction value listed in Table S6 [1].
- The uncertainty for
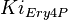 in HeLa cell line is mentioned in the forward reaction as
in HeLa cell line is mentioned in the forward reaction as 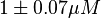 and in the reverse direction as
and in the reverse direction as 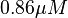 . The mean value is calcualated as
. The mean value is calcualated as 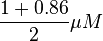 and the standard deviation is calculated based on the same ratio mentioned in forward reaction which comes as
and the standard deviation is calculated based on the same ratio mentioned in forward reaction which comes as 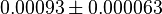 in mM.
in mM.
- Same principle is used for
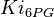 . The mean value is
. The mean value is 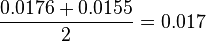 and the standard deviation is calculted based on the same ratio of reverse reaction for HeLa cell line; 0.0043
and the standard deviation is calculted based on the same ratio of reverse reaction for HeLa cell line; 0.0043
- There are two reported value for
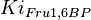 ; 0.1[3], 0.06[1]. The mean and standard deviation are calcualted from these two values.
; 0.1[3], 0.06[1]. The mean and standard deviation are calcualted from these two values.
| Parameter | Value | Units |
|---|---|---|

|
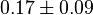
|

|
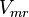
|
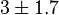
|

|
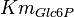
|
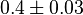 mM mM
|
mM |
|
|
mM |
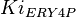
|
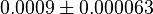
|
mM |
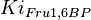
|
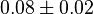
|
mM |
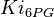
|
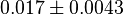
|
mM |
Equilibrium constant
| Equilibrium constant | Conditions | Source |
|---|---|---|
| 0.50 | pH=7, T=25°C | Lehninger, (2008)[4] p 553:
|
| 0.51 | pH=7, T=25°C | Lehninger, (1975)[5] p 396: Keq(reverse)=1.97 => Keq(forward)=1/1.97=0.51. |
| 0.41 | pH=7, T=25°C | Voet et al.[6] from Newshole et al. (1973) [7]p 97:
|
- Averaging these values gives
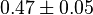
Refences
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et. al. (2011). Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 (doi) Cite error: Invalid
<ref>tag; name "Hernandez2011" defined multiple times with different content Cite error: Invalid<ref>tag; name "Hernandez2011" defined multiple times with different content - ↑ Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988(doi)
- ↑ Marin-Hernandez, A., Gallardo-Perez, J. C., Ralph, S. J., Rodriguez-Enriquez, S. & Moreno-Sanchez, R. (2009), HIF-1α modulates energy metabolism in cancer cells by inducing over-expression of specific glycolytic isoforms. Mini-Rev. Med. Chem. 9, 1084–1101
- ↑ David L. Nelson, Michael M. Cox (2008), Lehninger Principles of Biochemistry (5th edn), W. H. Freeman and Company
- ↑ Lehninger, A.L. (1975) Biochemistry (2nd edn), Worth
- ↑ Voet, D., Voet., J.G. and Pratt, C. W. (1999) Fundamentals of biochemistry, Wiley
- ↑ Newshole, E.A. and Stuart, C. (1973) Regulation in Metabolism, Wiley
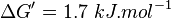 ,
, 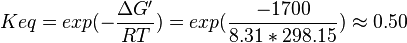
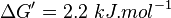 ,
,