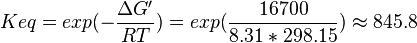Hexokinase
The enzyme hexokinase phosphorylates (adds a phosphate group to) glucose in the cell's cytoplasm. In the process, a phosphate group from Adenosine triphosphate (ATP) is transferred to glucose producing glucose 6-phosphate (Glc6P).
Contents
Chemical equation
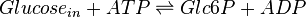
Rate equation
Without inhibition
Random Bi-Bi Michaelis Menten is used.[1]
![v = \frac{\frac{V_{mf}}{Km_{Glucose_{in}}K_{ATP}}\Big( [Glucose_{in}][ATP] - \frac{[Glc6P][ADP]}{K_{eq}} \Big)}{1 + \frac{[Glucose_{in}]}{Km_{Glucose_{in}}} + \frac{[ATP]}{K_{ATP}} + \frac{[Glucose_{in}][ATP]}{Km_{Glucose_{in}}K_{ATP}} + \frac{[Glc6P]}{K_{Glc6P}} + \frac{[ADP]}{K_{ADP}} +\frac{[Glc6P][ADP]}{K_{Glc6P}K_{ADP}} + \frac{[Glucose_{in}][ADP]}{K_{Glucose_{in}}K_{ADP}} +\frac{[Glc6P][ATP]}{K_{Glc6P}K_{ATP}} }](/wiki/images/math/d/2/d/d2d1c3d1115eef8cdf23e7eb98f65b04.png)
With allosteric inhibition
Hexokinase is allosterically inhibited by Glc6P [2]. The rate law taking into account this inhibition is
![\frac{V_{mf}* \frac{[Glucose]*[ATP]}{Km_{Glucose}*Km_{ATP}} }{ \left(1 + \frac{[Glc6P]}{Ki_{Glc6P}} \right)* \left( 1 + \frac{[Glucose]}{Km_{Glucose}} \right) + \frac{[ATP]}{Km_{ATP}} + \frac{[Glucose]*[ATP]}{Km_{Glucose}*Km_{ATP}} }](/wiki/images/math/c/2/6/c26962b0cefb2c26a75d64c2de18328e.png)
Parameters
Without inhibition
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.04 [3] | 
|
HeLa cell line | |

|
651[4] | |||
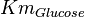
|
0.1[1] | mM | ||
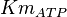
|
1.1[1] | mM | ||
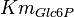
|
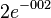 [1] [1]
|
mM | ||
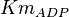
|
3.5[1] | mM |
With inhibition
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.04 [3] | 
|
HeLa cell line | |
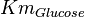
|
0.1[1] | mM | ||
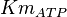
|
1.1[1] | mM | ||
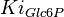
|
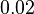 [2] [2]
|
mM |
Parameters with uncertainty
Hexokinase isoenzyme has been reported to vary in different developmental and metabolic status of the cell. In mammalian tissues four isoenzymes of Hexokinase is present [5]. Hexokinase-II has been found to be overexpressed in several first growith cancer cells [3]. Therefore in our model we have considered only the Hexokinase-II kinetic parameter values. The value of Hexokinase is reported as 0.02 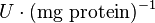 . Converting this value to
. Converting this value to 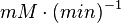 gives the value of 0.0013
gives the value of 0.0013 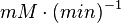 . But in the model file the authors used the value of 0.055 and we consider this value for our model as well. The ratio of error is the same as mentioned in the paper.
. But in the model file the authors used the value of 0.055 and we consider this value for our model as well. The ratio of error is the same as mentioned in the paper.
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
Failed to parse (Cannot store math image on filesystem.): 0.055 \pm 0.0165 (n=4)[6] | 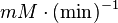
|
HeLA cell line | |
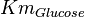
|
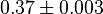 (n=5)[7] (n=5)[7]
|
mM | SNU449 cell (Human Liver Tissue) | |
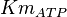
|
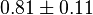 (n=5)[7] (n=5)[7]
|
mM | ||
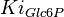
|
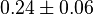 (n=5)[7] (n=5)[7]
|
mM |
Equilibrium constant
| Equilibrium constant | Conditions | Source |
|---|---|---|
| 4908 | pH=7, T=25°C | Voet et al.[8] from Newshole et al. (1973) [9]:
|
| 846 | pH=7, T=25°C | Lehninger, (2008)[10] p 553:
|
| 890 | pH=7, T=25°C | Lehninger, (1975)[11] p 396 |
- Calculating average from these equlibrium constant gives the value of
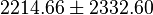
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011) Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 (doi)
- ↑ 2.0 2.1 Wilson J E (2003). Isozymes of mammalian hexokinase: structure, subcellular localization and metabolic function. Journal of Experimental Biology, 206, pp. 2049–2057 (doi)
- ↑ 3.0 3.1 3.2 Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988(doi) Cite error: Invalid
<ref>tag; name "Hernandez_2006" defined multiple times with different content - ↑ Arbitrary value
- ↑ Adams V, Kempf W, Hassam S, Briner J. (1995), Determination of hexokinase isoenzyme I and II by RT-PCR: increased hexokinase II isoenzyme in human renal cell carcinoma. J Biochem Mol Med 1995;54:53–58.
- ↑ Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988 (doi) (Model File)
- ↑ 7.0 7.1 7.2 Ahn, K.J.; Kim, J.; Yun, M.; Park, J.H.; Lee, J.D.(2009), Enzymatic properties of the N- and C-terminal halves of human hexokinase II, BMB Rep. 42, 350-355.
- ↑ Voet, D., Voet., J.G. and Pratt, C. W. (1999) Fundamentals of biochemistry, Wiley
- ↑ Newshole, E.A. and Stuart, C. (1973) Regulation in Metabolism, Wiley
- ↑ David L. Nelson, Michael M. Cox (2008), Lehninger Principles of Biochemistry (5th edn), W. H. Freeman and Company
- ↑ Lehninger, A.L. (1975) Biochemistry (2nd edn), Worth
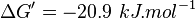 ,
, 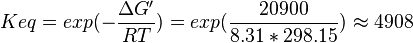
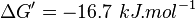 ,
,