Difference between revisions of "Degradation of R"
(→Parameters) |
(→Parameters) |
||
| Line 25: | Line 25: | ||
|- | |- | ||
|<math>d_{R}</math> | |<math>d_{R}</math> | ||
| − | |<math> | + | |<math>5.02 \cdot 10^{-4}- 0.00578 min^{-1}</math> <ref name="Trötschel2013"> [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3815937/pdf/mbt0006-0708.pdf Trötschel C, Albaum SP, Poetsch A. ''Proteome turnover in bacteria: current status for Corynebacterium glutamicum and related bacteria.'' Microbial biotechnology. 2013;6(6):708-719.]</ref> <ref name="Jayapal2010"> [http://pubs.acs.org/doi/pdf/10.1021/pr9007738 Jayapal KP, Sui S, Philp RJ, Kok YJ, Yap MG, Griffin TJ, Hu WS. ''Multitagging proteomic strategy to estimate protein turnover rates in dynamic systems.'' J Proteome Res. 2010 May 7;9(5):2087-97.]</ref> <ref name="Mosteller1980"> [http://www.jbc.org/content/255/6/2524.full.pdf Mosteller RD, Goldstein RV, Nishimoto KR. ''Metabolism of individual proteins in exponentially growing Escherichia coli.'' J Biol Chem. 1980 Mar 25;255(6):2524-32]</ref> |
|<math> min^{-1} </math> | |<math> min^{-1} </math> | ||
|N/A | |N/A | ||
Revision as of 01:01, 23 September 2017
The ScbR monomer (R) degrades.
Contents
Chemical equation
Rate equation
![r= d_{R}\cdot[R]](/wiki/images/math/6/7/5/675d245d387b813d8bcaf71d7a91c6f9.png)
Parameters
The parameter of this reaction is the degradation rate of R (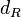 ). The parameter values were derived from proteomics studies on different proteins of S. coelicolor.
). The parameter values were derived from proteomics studies on different proteins of S. coelicolor.
| Name | Value | Units | Value in previous GBL models [1] [2] | Remarks-Reference |
|---|---|---|---|---|
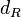
|
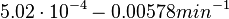 [3] [4] [5] [3] [4] [5]
|

|
N/A | In a quantitative proteomics study on protein turnover rates in dynamic systems, Jayapal et al. reported the degradation rates of 115 proteins in S. coelicolor cultures undergoing transition from exponential growth to stationary phase. The values were in the range 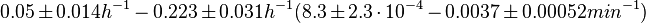 with a median of with a median of 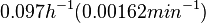 . .
 Jayapal et al. 2012[4] The full list of the proteins studied and their turnover rate constants can be found here. |
Parameters with uncertainty
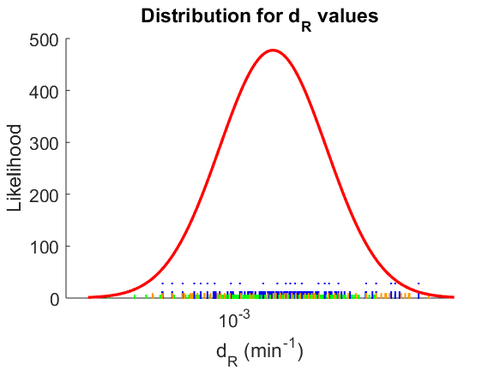
When deciding how to describe the uncertainty for this parameter we must take into consideration that the reported values are estimated or calculated from proteomics experiments, using methods which were prone to measurement errors (i.e. optical density of fragmented mycelia to estimate the growth rate (μ)) and extrapolations from cell density to total cellular protein without taking into account the dilution due to cell division. Additionally, the study did not include measurements on ScbR protein specifically, although it was conducted on a wide range of S. coelicolor proteins which enables a rough estimation of the range of values of protein degradation in this species. These facts influence the quantification of the parameter uncertainty and therefore the shape of the corresponding distribution. By assigning the appropriate weights to the parameter values and using the method described here, the appropriate probability distribution was designed.
The mode of the log-normal distribution of the 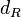 to
to 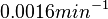 . In order to explore the full range of published values and even sample a percentage of values outside the reported range, the confidence interval factor is set to
. In order to explore the full range of published values and even sample a percentage of values outside the reported range, the confidence interval factor is set to 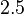 , so that the range where 95.45% of the values are found is between
, so that the range where 95.45% of the values are found is between 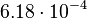 and
and 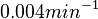 .
.
The probability distribution for the parameter, adjusted accordingly in order to reflect the above values, is the following:
The location and scale parameters of the distribution are:
| Parameter | μ | σ |
|---|---|---|
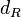
|
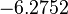
|
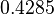
|
References
- ↑ S. Mehra, S. Charaniya, E. Takano, and W.-S. Hu. A bistable gene switch for antibiotic biosynthesis: The butyrolactone regulon in streptomyces coelicolor. PLoS ONE, 3(7), 2008.
- ↑ A. Chatterjee, L. Drews, S. Mehra, E. Takano, Y.N. Kaznessis, and W.-S. Hu. Convergent transcription in the butyrolactone regulon in streptomyces coelicolor confers a bistable genetic switch for antibiotic biosynthesis. PLoS ONE, 6(7), 2011.
- ↑ Trötschel C, Albaum SP, Poetsch A. Proteome turnover in bacteria: current status for Corynebacterium glutamicum and related bacteria. Microbial biotechnology. 2013;6(6):708-719.
- ↑ 4.0 4.1 Jayapal KP, Sui S, Philp RJ, Kok YJ, Yap MG, Griffin TJ, Hu WS. Multitagging proteomic strategy to estimate protein turnover rates in dynamic systems. J Proteome Res. 2010 May 7;9(5):2087-97.
- ↑ Mosteller RD, Goldstein RV, Nishimoto KR. Metabolism of individual proteins in exponentially growing Escherichia coli. J Biol Chem. 1980 Mar 25;255(6):2524-32