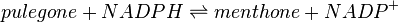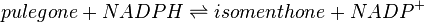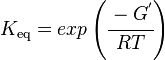Double-bond reductase (DBR)
You can go back to main page of the kinetic model here.
Legend:
| Have not started ·· | 1 -2 data found ·· | 3-4 data found ·· | sufficient data found/estimated ·· | data distribution generated ·· | data sampled |
| to do | DONE! |
Contents
What we know
Pulegone reductase(s) (PGR) catalyses the NADPH-dependent convertion of pulegone to menthone and isomenthone (the former predominates).
Issues
Strategies
Reaction catalysed
File:DBR_reaction_diagram_v1.png
Enzyme and Metabolite Background Information
Long metabolite names are abbreviated in the model for clarity and standard identification purposes.
| Metabolite | Abbreviation | Chemical Formula | Molar mass (g/mol) | ChEBI | ChEMBL | PubChem | BRENDA | PlantCyc |
|---|---|---|---|---|---|---|---|---|
| pulegone reductase | PGR | 37914 Da | 1.3.1.81 | |||||
| pulegone | C10H16O | 136.24 | ||||||
| menthone | ||||||||
| NADPH | C21H30N7O17P3 | 745.42116 | 16474 | |||||
| NADP+ | C21H29N7O17P3 | 744.41322 | 18009 | |||||
| isomenthone |
Equation Rate
Two PGR reactions are included in the kinetic model with each converting pulegone to methone and isomenthone respectively. Both reactions are parameterised using random Bi-Bi reversible Michaelis-Menten equation.
Reaction 1: Conversion of pulegone to menthone
- Failed to parse (Cannot store math image on filesystem.): V_\mathrm{PGR} = Kcat_\mathrm{forward} * [PGR] * \cfrac {\left ( \cfrac{[pulegone]}{Km_\mathrm{pulegone}} * \cfrac {[NADPH]}{Km_\mathrm{NADPH}} \right ) * \left ( 1 - \cfrac {[menthone]*[NADP]}{[pulegone]*[NADPH]*K_\mathrm{eq}} \right )} { \left (1 + \cfrac {[NADPH]}{Km_\mathrm{NADPH}} + \cfrac {[NADP]}{Km_\mathrm{NADP}} \right ) + \left ( 1+ \cfrac {[pulegone]}{Km_\mathrm{pulegone}} + \cfrac {[menthone]}{Km_\mathrm{menthone}} \right ) }
Reaction 2: Conversion of pulegone to isomenthone
- Failed to parse (Cannot store math image on filesystem.): V_\mathrm{PGR} = Kcat_\mathrm{forward} * [PGR] * \cfrac {\left ( \cfrac{[pulegone]}{Km_\mathrm{pulegone}} * \cfrac {[NADPH]}{Km_\mathrm{NADPH}} \right ) * \left ( 1 - \cfrac {[isomenthone]*[NADP]}{[pulegone]*[NADPH]*K_\mathrm{eq}} \right )} { \left (1 + \cfrac {[NADPH]}{Km_\mathrm{NADPH}} + \cfrac {[NADP]}{Km_\mathrm{NADP}} \right ) + \left ( 1+ \cfrac {[pulegone]}{Km_\mathrm{pulegone}} + \cfrac {[isomenthone]}{Km_\mathrm{isomenthone}} \right ) }
| Parameter | Description | Units |
|---|---|---|
| VPGR | Reaction rate for Limonene-3-hydroxylase | μM/min |
| Kcatforward | Catalytic constant in the forward direction | s-1 |
| Kmpulegone | Michaelis-Menten constant for pulegone | μM |
| Kmmenthone | Michaelis-Menten constant for menthone | μM |
| Kmisomenthone | Michaelis-Menten constant for isomenthone | μM |
| KmNADPH | Michaelis-Menten constant for NADPH | μM |
| KmNADP | Michaelis-Menten constant for NADP+ | μM |
| Keq | Equilibrium constant | |
| [PGR] | enzyme concentration | μM |
| [pulegone] | Pulegone concentration | μM |
| [menthone] | Menthone concentration | μM |
| [isomenthone] | Isomenthone concentration | μM |
| [NADPH] | NADPH concentration | μM |
| [NADP] | NADP+ concentration | μM |
Strategies for estimating the kinetic parameter values
Standard Gibbs Free energy
The Gibbs free energy for PGR is -3.9565125 kcal.mol^-1. This value is estimated from the 'Contribution group' method by Latendresse, M. and is available from MetaCyc (EC 1.3.1.81) [1].
Calculating the Equilibrium Constant
The equilibrium constant can be calculated using the Van't Hoff Isotherm equation:
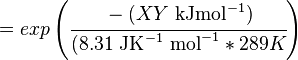
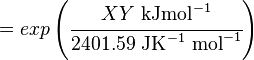
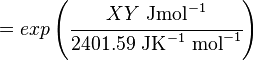
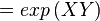
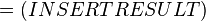
where;
| Keq | Equilibrium constant |
| -ΔG° | Gibbs free energy change. For (INSERT ENZYME) it is (INSERT VALUE) kJmol-1 |
| R | Gas constant with a value of 8.31 JK-1mol-1 |
| T | Temperature which is always expressed in kelvin |
Extracting Information from menthone Production Rates
A table will go here
Published Kinetic Parameter Values
Km Values
| Parameter | Directionality | Substrate / Product | Value | unit | Method notes | References |
|---|---|---|---|---|---|---|
| Km | Forward | pulegone | 2.3 | µM | Gene from peppermint oil gland secretory cell cDNA, expressed in E. coli, optimal pH 5.0, menthone:isomenthone ratio of 55:45 | Ringer2003 |
| Km | Forward | pulegone | 2.9 | µM | Gene from peppermint oil gland secretory cell cDNA, expressed in E. coli, optimal pH 5.0, menthone:isomenthone ratio of 55:45, Km 2.3 +/- 0.6 | Ringer2003 |
| Km | Forward | NADPH | 6.9 | µM | Gene from peppermint oil gland secretory cell cDNA, expressed in E. coli, optimal pH 5.0, menthone:isomenthone ratio of 55:45 | Ringer2003 |
Detailed description of kinetic values obtained from literature
A more detailed description of the values listed above can be found here .
Simulations
References
- ↑ Latendresse M. (2013). "Computing Gibbs Free Energy of Compounds and Reactions in MetaCyc."