Difference between revisions of "DXS"
Aliah.hawari (talk | contribs) (→DXS parameters) |
Aliah.hawari (talk | contribs) (→DXS parameters) |
||
| Line 71: | Line 71: | ||
|} | |} | ||
| − | [[File: | + | [[File:DXS_KmG3p.png | left |600px ]] |
{|class='wikitable' | {|class='wikitable' | ||
Revision as of 15:08, 23 March 2017
You can go back to main page of the kinetic model here.
The DXS reaction (EC 2.2.1.7)
Deoxyxylulose-5-phosphate synthase (DXS) catalyses the production of 1-deoxy-D-xylulose 5-phosphate (DXP) from pyruvate and glyceraldehyde 3-phosphate (G3P). This reaction is the first step in the MEP pathway.
Modelling DXS
In the kinetic model, the DXS reaction is modelled with reversible Michaelis-Menten using the Hanekom [1] bi-bi random order generic equation. In total, this reaction requires five kinetic parameters (Kms for all substrates and products, and a forward Kcat) and one thermodynamic parameter (Equilibrium constant, Keq).
DXS parameters
| Parameter | Direction | Substrate | Value | Unit | Weight | Description | Reference |
|---|---|---|---|---|---|---|---|
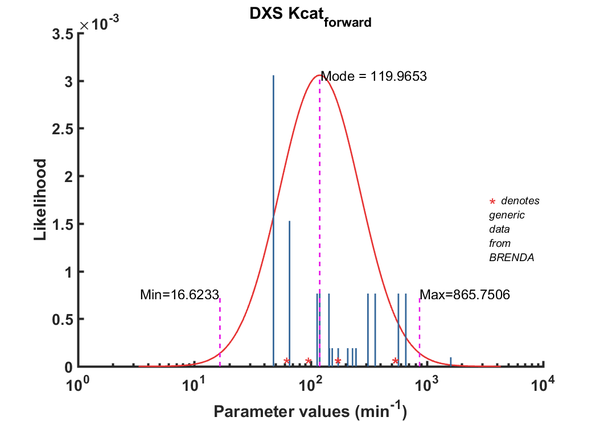
| Kcat | Forward | DXS | 229.7 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 40mM Tris, pH 8, 37C ; Km_GAP:52.5 +/- 8.3 microM; Km_pyruvate: 86.3 +/- 16.2 microM, kcat: 145.50 +/- 12.7 1/min | [2] |
| Kcat | Forward | DXS | 153.6 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 40mM Tris, pH 8, 37C ; Km_GAP:52.5 +/- 8.3 microM; Km_pyruvate: 86.3 +/- 16.2 microM, kcat: 145.50 +/- 12.7 1/min | [2] |
| Kcat | Forward | DXS | 145.5 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 40mM Tris, pH 8, 37C ; Km_GAP:52.5 +/- 8.3 microM; Km_pyruvate: 86.3 +/- 16.2 microM, kcat: 145.50 +/- 12.7 1/min | [2] |
| Kcat | Forward | DXS | 209 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 100mM Tris, pH 8, 37C ; Km_GAP:279.0+/- 7.2microM; Km_pyruvate: 74.70+/- 7.3 microM, kcat: 209.0 +/- 6.3 1/min | [2] |
| Kcat | Forward | DXS | 173 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 100mM Tris, pH 8, 37C ; Km_GAP:279.0+/- 7.2microM; Km_pyruvate: 74.70+/- 7.3 microM, kcat: 209.0 +/- 6.3 1/min | [2] |
| Kcat | Forward | DXS | 246 | 1/min | 16 | from E. coli wild type DXS, with non-optimal buffer: 100mM Tris, pH 8, 37C ; Km_GAP:279.0+/- 7.2microM; Km_pyruvate: 74.70+/- 7.3 microM, kcat: 209.0 +/- 6.3 1/min | [2] |
| Kcat | Forward | GAP | 48 | 1/min | 256 | Taken from Cane 2001's ref20. E.coli DXS in 40mM Tris, pH7.5, 37¡C. Km pyruvate 2.9 ± 0.5 mM. | [3] |
| Kcat | Forward | GAP | 66 | 1/min | 128 | DXPS2; in vitro- S. coelicolor gene expressed in E. coli; pH 7.5, 47C. | [4] |
| Kcat | Forward | GAP | 114 | 1/min | 8 | from R. capsulatus, pH 7.4, 37C | [5] |
| Kcat | Forward | GAP | 660 | 1/min | 64 | from Plasmodium, expressed in E. coli. Look at Table 3. pH7-7.5;37C, Km_GAp:19 +/- 4 microM; Km_Pyruvate: 870 +/- 110 microM. | [6] |
| Kcat | Forward | GAP | 1608 | 1/min | 8 | dxs11 from Agrobacterium tumefaciens, pH8.0, 37¡C, expressed in E. coli | [7] |
| Kcat | Forward | GAP | 120 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Kcat | Forward | GAP | 120 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Kcat | Forward | GAP | 360 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Kcat | Forward | Pyruvate | 570 | 1/min | 64 | from Plasmodium, expressed in E. coli. Look at Table 3. pH7-7.5;37C, Km_GAp:19 +/- 4 microM; Km_Pyruvate: 870 +/- 110 microM. | [6] |
| Kcat | Forward | Pyruvate | 144 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Kcat | Forward | Pyruvate | 114 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Kcat | Forward | Pyruvate | 312 | 1/min | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Parameter | Direction | Substrate | Value | Unit | Weight | Description | Reference |
|---|---|---|---|---|---|---|---|
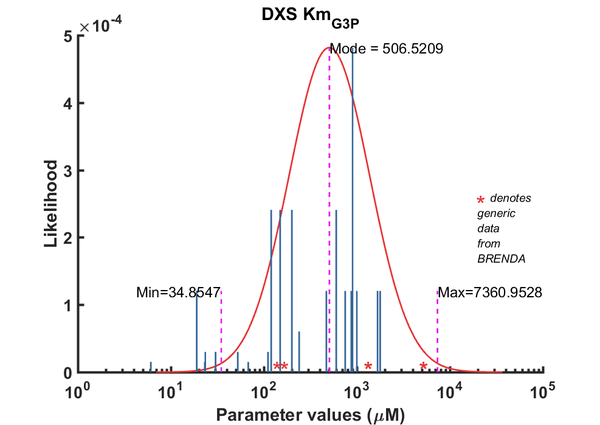
| Km | Forward | G3P | 6.1 | µM | 8 | pH 7.9, 37C (optimal), Mycobacterium tuberculosis, Km_G3P 6.1 microM, Km_Pyr 40 microM | Bailey2002, Br658834 |
| Km | Forward | G3P | 1670 | µM | 64 | from Vitis vinifera L. ; expressed in E. coli DH5a, pH 8.0; 37C; VvDXS N284 | Battilana2011, Br720131 |
| Km | Forward | G3P | 1780 | µM | 64 | from Vitis vinifera L. ; expressed in E. coli DH5a, pH 8.0; 37C; VvDXS N284 | Battilana2011, Br720131 |
| Km | Forward | G3P | 900 | µM | 256 | Taken from Cane 2001's ref20. E.coli DXS in 40mM Tris, pH7.5, 37¡C. Km pyruvate 2.9 ± 0.5 mM. | Boronat1999 |
| Km | Forward | G3P | 750 | µM | 64 | from Capsicum anuum; Km_pyruvate: 500 microM; Km_GAP: 750 microM | Bouvier1998 |
| Km | Forward | G3P | 30.3 | µM | 16 | from E. coli wild type DXS, Optimal buffer: 100 mM HEPES, pH 8, 37C ; Km_GAP:23.5 +/- 1.7 microM; Km_pyruvate: 48.7 +/- 8.0 microM, kcat: 153.6 +/- 6.8 1/min | [2] |
| Km | Forward | G3P | 23.5 | µM | 16 | from E. coli wild type DXS, Optimal buffer: 100 mM HEPES, pH 8, 37C ; Km_GAP:23.5 +/- 1.7 microM; Km_pyruvate: 48.7 +/- 8.0 microM, kcat: 153.6 +/- 6.8 1/min. Kinetics were measured in different buffers (buffer optimization study). All buffers ph*, 37C | [2] |
| Km | Forward | G3P | 52.5 | µM | 16 | from E. coli wild type DXS, Optimal buffer: 100 mM HEPES, pH 8, 37C ; Km_GAP:23.5 +/- 1.7 microM; Km_pyruvate: 48.7 +/- 8.0 microM, kcat: 153.6 +/- 6.8 1/min | [2] |
| Km | Forward | G3P | 600 | µM | 128 | DXPS1; in vitro- S. coelicolor gene expressed in E. coli; pH 7.5, 47C. | [4] |
| Km | Forward | G3P | 200 | µM | 128 | DXPS2; in vitro- S. coelicolor gene expressed in E. coli; pH 7.5, 47C. | [4] |
| Km | Forward | G3P | 68 | µM | 8 | from Rhodobacter capsulatus, pH7.4, 37C, Km_GAP: 0.068 +/- 0.001 mM, Km_pyr: 0.44 +/- 0.05 mM. | [5] |
| Km | Forward | G3P | 111 | µM | 16 | from Grey poplar leaves, pH 8.6; 45 C; | Ghirardo2010 |
| Km | Forward | G3P | 150 | µM | 128 | from Rhodobacter capsulatus, pH7.4, 37¡C; expressed in E. coli; recombinant isoenzyme-A; Km_pyruvate: 610 +/- 50 microM; Km_GAP: 150 +/- 10 microM; Vmax = 1.9 +/- 0.1 micrommol/min/mg; | Hahn2001, Br395816 |
| Km | Forward | G3P | 120 | µM | 128 | from Rhodobacter capsulatus, pH7.4, 37¡C; expressed in E. coli; recombinant isoenzyme-A; Km_pyruvate: 610 +/- 50 microM; Km_GAP: 150 +/- 10 microM; Vmax = 1.9 +/- 0.1 micrommol/min/mg; | Hahn2001, Br395816 |
| Km | Forward | G3P | 19 | µM | 64 | from Plasmodium, expressed in E. coli. Look at Table 3. pH7-7.5;37C, Km_GAp:19 +/- 4 microM; Km_Pyruvate: 870 +/- 110 microM. | [6] |
| Km | Forward | G3P | 120 | µM | 8 | from Streptomyces sp. strain CL190, expressed in E. coli. pH 9.0, 42-44C. Table 1. Km_pyruvate: 65microM, Km_GAP: 120 microM | Kuzuyama2000, Br395812 |
| Km | Forward | G3P | 240 | µM | 32 | from E. coli. pH 7.5-8.0.0, 42-44C. Km_pyruvate: 65microM, Km_GAP: 120 microM. See Table 1. Measured to compare with DXS from streptomyces. Purified in Kuzuyama1998 study. | Kuzuyama2000, Br395812 |
| Km | Forward | G3P | 23.2 | µM | 8 | dxs11 from Agrobacterium tumefaciens, pH8.0, 37¡C, expressed in E. coli | Lee2007, Br674982 |
| Km | Forward | G3P | 470 | µM | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Km | Forward | G3P | 870 | µM | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
| Km | Forward | G3P | 1000 | µM | 64 | from Botrycoccus braunnii. Three recombinant enzymes used: DXS-I, DXS-II, DXS-III which are different by the digestion pattern using Xhol and BamHI. expressed in E. coli; pH 7.8 , 32 C; Km 1800 +/- 200 microM | [8] |
References
- ↑ Hanekom, A. J. 2006. "Generic kinetic equations for modelling multisubstrate reactions in computational systems biology", MSc Thesis submitted at the University of Stellenbosch
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 Brammer, L.A. 2011 "1-FDeoxy-D-xylulose 5-phosphate synthase catalyzes a novel random sequential mechanism", JBioChem, 283(42):36522-36531.
- ↑ Brammer, L.A. 2011 "1-Deoxy-D-xylulose 5-phosphate synthase catalyzes a novel random sequential mechanism", JBioChem, 283(42):36522-36531.
- ↑ 4.0 4.1 4.2 Cane, D.E., et. al.. 2001 "Molecular cloning, expression and characterization of the first three genes in the mevalonate-independent isoprenoid pathway in Streptomyces coelicolor ", Bioorganic & Medicinal Chemistry, 9:1467-1477.
- ↑ 5.0 5.1 Eubanks, L.M. & Poulter, C.D. 2003. "Rhodobacter capsulatus 1-Deoxy-D-xylulose 5-Phosphate Synthase: Steady-State Kinetics and Substrate Binding† ", Biochemistry, 42:1140-1149.
- ↑ 6.0 6.1 6.2 Handa, S. et. al. 2013. "Production of recombinant 1-deoxy-d-xylulose-5-phosphate synthase from Plasmodium vivax in Escherichia coli", Biochemistry, 3:124-129. Cite error: Invalid
<ref>tag; name "Handa2013" defined multiple times with different content - ↑ Lee, J. et. al. 2007. "Cloning and characterization of the dxs gene, encoding 1-deoxy-d-xylulose 5-phosphate synthase from Agrobacterium tumefaciens, and its overexpression in Agrobacterium tumefaciens", JBiotech, 128:555-566.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 Matsushima, D. et. al2012. "The single cellular green microalga Botryococcus braunii, race B possesses three distinct 1-deoxy-d-xylulose 5-phosphate synthases", PlantSci, 185-186:309-320.
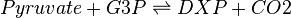
![V_\mathrm{DXS}= \cfrac{Kcat_\mathrm{forward} \bullet [DXS] \bullet \left( \cfrac{[Pyr]}{Km_\mathrm{DXS}} \right) \bullet \left( \cfrac{[G3P]}{Km_\mathrm{g3p}} \right) \bullet \left( 1 - \cfrac{\left( \cfrac{[DXP]\bullet[CO2]}{[Pyr]\bullet[G3P]} \right)}{K_\mathrm{eq}} \right)} {\left( 1 + \cfrac {[Pyr]}{Km_\mathrm{pyr}} + \cfrac{[CO2]}{Km_\mathrm{co2}}\right) \bullet \left( 1 + \cfrac{[G3P]}{Km_\mathrm{g3p}} + \cfrac{[DXP]}{KM_\mathrm{dxp}} \right)}](/wiki/images/math/4/a/8/4a8faa589ce739bde2b441190759da1b.png)