Difference between revisions of "Monocarboxylate (Lactate) transporter"
| Line 7: | Line 7: | ||
Reversible Michaelis-Menten rate law is used | Reversible Michaelis-Menten rate law is used | ||
| − | <center><math> \frac{V_{f}\frac{[Lactate_{in}]}{Km_{Lactate_{in}} - out}}{} </math></center> | + | <center><math> \frac{V_{f}\frac{[Lactate_{in}]}{Km_{Lactate_{in}}}- V_{r}\frac{[Lactate_{out}]}{Km_{Lactate_{out}}}}{1+ \frac{[Lactate_{in}]}{Km_{Lactate_{in}}} - \frac{[Lactate_{out}]}{Km_{Lactate_{out}}} } </math></center> |
==References== | ==References== | ||
<references/> | <references/> | ||
Revision as of 11:35, 28 February 2014
Monocarboxylate transporters,[1] or MCTs, constitute a family of proton-linked plasma membrane transporters that carry molecules having one carboxylate group (monocarboxylates), such as lactate and pyruvate.
Chemical equation
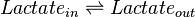
Rate equation
Reversible Michaelis-Menten rate law is used
![\frac{V_{f}\frac{[Lactate_{in}]}{Km_{Lactate_{in}}}- V_{r}\frac{[Lactate_{out}]}{Km_{Lactate_{out}}}}{1+ \frac{[Lactate_{in}]}{Km_{Lactate_{in}}} - \frac{[Lactate_{out}]}{Km_{Lactate_{out}}} }](/wiki/images/math/a/b/c/abc535a21e702a99833c028802c5fcf3.png)