Difference between revisions of "Glyceraldehyde-3-phosphate dehydrogenase"
| Line 5: | Line 5: | ||
== Rate equation == | == Rate equation == | ||
| + | |||
| + | Ordererd Ter-Bi reversible Michaelis-Menten equation for non-interacting substrates. <ref name="Hernandez2011"> Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011) Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 ([http://dx.doi.org/10.1016/j.bbabio.2010.11.006 doi]) </ref> | ||
| + | |||
<center><math>v = \frac{ V_{mf}\frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} - V_{mr} \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} }{1 + \frac{[NAD]}{K_{NAD}} + \frac{[NAD][Gly3P]}{K_{NAD}K_{Gly3P}} + \frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} + \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} + +\frac{[NADH]}{K_{NADH}} }</math></center> | <center><math>v = \frac{ V_{mf}\frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} - V_{mr} \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} }{1 + \frac{[NAD]}{K_{NAD}} + \frac{[NAD][Gly3P]}{K_{NAD}K_{Gly3P}} + \frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} + \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} + +\frac{[NADH]}{K_{NADH}} }</math></center> | ||
== Parameters == | == Parameters == | ||
== Alternate parameter values == | == Alternate parameter values == | ||
| + | ==References== | ||
| + | <referenfces/> | ||
Revision as of 15:26, 27 February 2014
This enzyme serves two functions in this step. First the enzyme transfers a hydrogen (H-) from glyceraldehyde phosphate to the oxidizing agent nicotinamide adenine dinucleotide (NAD+) to form NADH. Next it adds a phosphate (P) from the cytosol to the oxidized glyceraldehyde phosphate to form 1, 3-bisphosphoglycerate.
Chemical equation
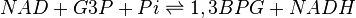
Rate equation
Ordererd Ter-Bi reversible Michaelis-Menten equation for non-interacting substrates. [1]
![v = \frac{ V_{mf}\frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} - V_{mr} \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} }{1 + \frac{[NAD]}{K_{NAD}} + \frac{[NAD][Gly3P]}{K_{NAD}K_{Gly3P}} + \frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} + \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} + +\frac{[NADH]}{K_{NADH}} }](/wiki/images/math/c/e/c/cec09052887411fec63867405d65296f.png)