Difference between revisions of "Phosphoglycerate dehydrogenase"
(→Parameters with uncertainty) |
|||
| Line 42: | Line 42: | ||
==Parameters with uncertainty== | ==Parameters with uncertainty== | ||
| − | *serA is the enzyme concentration collected from Turnaev 'et. al.' <ref name="Turnaev_2006"></ref> which is considered to be fixed. | + | *serA is the enzyme concentration collected from Turnaev ''et. al.'' <ref name="Turnaev_2006"></ref> which is considered to be fixed. |
{|class="wikitable" | {|class="wikitable" | ||
| Line 73: | Line 73: | ||
| mM | | mM | ||
|} | |} | ||
| − | |||
==References== | ==References== | ||
<references/> | <references/> | ||
Revision as of 13:07, 6 May 2014
The enzyme Phosphoglycerate dehydrogenase (PDH) catalyzes the reaction to convert 3-phospho-D-glycerate (P3G) to phosphonooxypyruvate (PHP). The cofactors NADH and NAD+.
Contents
Chemical equation
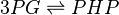
Rate equation
Modified rate equation is used [1]
Parameters
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|
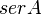
|
1.15 [2] | mM | Escherichia coli | |
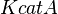
|
0.55 [3] | 1/s | ||
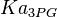
|
1.2[3] | mM | ||
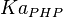
|
0.0032[3] | mM | ||
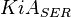
|
0.0038[3] | mM |
Parameters with uncertainty
- serA is the enzyme concentration collected from Turnaev et. al. [2] which is considered to be fixed.
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|
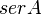
|
1.15 [2] | mM | Escherichia coli | |
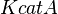
|
0.55 [3] | 1/s | ||
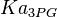
|
1.2[3] | mM | ||
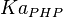
|
0.0032[3] | mM | ||
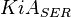
|
0.0038[3] | mM |
References
- ↑ Smallbone K, Stanford NJ (2013). Kinetic modeling of metabolic pathways: Application to serine biosynthesis. In: Systems Metabolic Engineering, Humana Press. pp. 113–121
- ↑ 2.0 2.1 2.2 Turnaev II, Ibragimova SS, Usuda Y et al (2006). Mathematical modeling of serine and glycine synthesis regulation in Escherichia coli. Proceedings of the fifth international conference on bioinformatics of genome regulation and structure 2:78–83
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 Zhao G, Winkler ME (1996). A novel alphaketoglutarate reductase activity of the serA-encoded 3-phosphoglycerate dehydrogenase of Escherichia coli K-12 and its possible implications for human 2-hydroxyglutaric aciduria. J Bacteriol 178:232–239