Difference between revisions of "Glyceraldehyde-3-phosphate dehydrogenase"
(→Parameters with uncertainty) |
(→Parameters with uncertainty) |
||
| Line 80: | Line 80: | ||
|- | |- | ||
|<math>Km_{1,3BPG}</math> | |<math>Km_{1,3BPG}</math> | ||
| − | |<math>0.013 \pm 0.0035</math | + | |<math>0.013 \pm 0.0035</math> |
|mM | |mM | ||
| | | | ||
|- | |- | ||
|<math>Km_{NAD+}</math> | |<math>Km_{NAD+}</math> | ||
| − | |<math>0.019 \pm 0.007</math | + | |<math>0.019 \pm 0.007</math> |
|mM | |mM | ||
| | | | ||
Revision as of 11:31, 29 April 2014
The Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) enzyme serves two functions in glycolytic pathway. First the enzyme transfers a hydrogen (H-) from glyceraldehyde phosphate (Gly3P) to the oxidizing agent Nicotinamide Adenine Dinucleotide (NAD+) to form NADH. Next it adds a phosphate (P) from the cytosol to the oxidized Gly3P to form 1, 3-bisphosphoglycerate.
Contents
Chemical equation
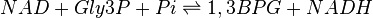
Rate equation
Ordererd Ter-Bi reversible Michaelis-Menten equation for non-interacting substrates. [1]
![v = \frac{ V_{mf}\frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} - V_{mr} \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} }{1 + \frac{[NAD]}{K_{NAD}} + \frac{[NAD][Gly3P]}{K_{NAD}K_{Gly3P}} + \frac{[NAD][Gly3P][Pi]}{K_{NAD}K_{Gly3P}K_{Pi}} + \frac{[1,3BPG][NADH]}{K_{1,3BPG}K_{NADH}} + +\frac{[NADH]}{K_{NADH}} }](/wiki/images/math/c/e/c/cec09052887411fec63867405d65296f.png)
Parameters
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.58 [2] | 
|
HeLa cell line | |
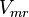
|
0.72[2] | 
| ||
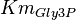
|
0.19[1] | mM | ||
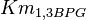
|
0.022[1] | mM | ||
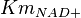
|
0.09[1] | mM | ||
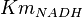
|
0.01[1] | mM | ||
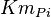
|
29[1] | mM |
Parameters with uncertainty
- Four values of
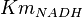 have been collected from Lambeir et. al. (1991)[3] for different organisms.
have been collected from Lambeir et. al. (1991)[3] for different organisms. 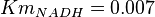 for Trypanosoma brucei,
for Trypanosoma brucei, 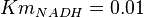 for Homo sapiens,
for Homo sapiens, 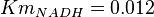 for Geobacillus stearothermophilus and
for Geobacillus stearothermophilus and 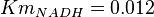 for Oryctolagus cuniculus. Calculating mean and std. dev. from these 4 values gives
for Oryctolagus cuniculus. Calculating mean and std. dev. from these 4 values gives 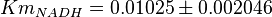
- Three values collected under different conditions from human cell is reported for
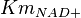 in Ryzlak (1998) et. al. [4]. Those values are 0.01, 0.02, 0.027. The mean and Std. Dev. from these three values are
in Ryzlak (1998) et. al. [4]. Those values are 0.01, 0.02, 0.027. The mean and Std. Dev. from these three values are 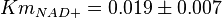
- Three values for
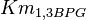 have also been reported under different conditions from human cell in Ryzlak (1998) et. al. [4]. Those values are 0.01, 0.012, 0.018. The mean and Std. Dev. from these three values are
have also been reported under different conditions from human cell in Ryzlak (1998) et. al. [4]. Those values are 0.01, 0.012, 0.018. The mean and Std. Dev. from these three values are 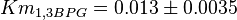
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
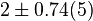 [2] [2]
|

|
HeLa cell line | |
|
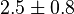 (5)[2] (5)[2]
|

|
||
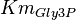
|
0.19[1] | mM | ||
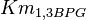
|
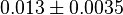
|
mM | ||
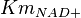
|
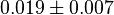
|
mM | ||
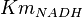
|
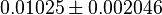
|
mM | Multiple organism | |
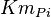
|
29[1] | mM |
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011) Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 (doi)
- ↑ 2.0 2.1 2.2 2.3 Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988(doi)
- ↑ Lambeir, A.M.; Loiseau, A.M.; Kuntz, D.A.; Vellieux, F.M.; Michels, P.A.M.; Opperdoes, F.R. (1991), The cytosolic and glycosomal glyceraldehyde-3-phosphate dehydrogenase from Trypanosoma brucei. Kinetic properties and comparison with homologous enzymes, Eur. J. Biochem. 198, 429-435
- ↑ 4.0 4.1 Ryzlak, M.T.; Pietruszko, R. (1998), Heterogeneity of glyceraldehyde-3-phosphate dehydrogenase from human brain, Biochim. Biophys. Acta 954, 309-324