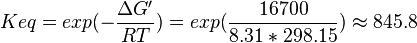Difference between revisions of "Hexokinase"
(→Parameters with uncertainty) |
|||
| (2 intermediate revisions by the same user not shown) | |||
| Line 80: | Line 80: | ||
== Parameters with uncertainty == | == Parameters with uncertainty == | ||
[[Category:Uncertainty]] | [[Category:Uncertainty]] | ||
| − | Hexokinase isoenzyme has been reported to vary in different developmental and metabolic status of the cell. In mammalian tissues four isoenzymes of Hexokinase is present <ref name="Adams_1995">Adams V, Kempf W, Hassam S, Briner J. (1995), ''Determination of hexokinase isoenzyme I and II by RT-PCR: increased hexokinase II isoenzyme in human renal cell carcinoma.'' J Biochem Mol Med 1995;54:53–58.</ref>. Hexokinase-II has been found to be overexpressed in several first growith cancer cells <ref name="Hernandez_2006"></ref>. Therefore in our model we have considered only the Hexokinase-II kinetic parameter values. | + | Hexokinase isoenzyme has been reported to vary in different developmental and metabolic status of the cell. In mammalian tissues four isoenzymes of Hexokinase is present <ref name="Adams_1995">Adams V, Kempf W, Hassam S, Briner J. (1995), ''Determination of hexokinase isoenzyme I and II by RT-PCR: increased hexokinase II isoenzyme in human renal cell carcinoma.'' J Biochem Mol Med 1995;54:53–58.</ref>. Hexokinase-II has been found to be overexpressed in several first growith cancer cells <ref name="Hernandez_2006"></ref>. Therefore in our model we have considered only the Hexokinase-II kinetic parameter values. The value of Hexokinase is reported as 0.06 <math>U\cdot(\text{mg protein})^{-1}</math>. Converting this value to <math>mM\cdot(min)^{-1}</math> gives the value of 3.9 <math>mM\cdot(min)^{-1}</math>. But in the model file the authors used the value of <math>0.04 \pm 0.01</math>. Same error ratio is used in our model. |
{|class="wikitable" | {|class="wikitable" | ||
| Line 90: | Line 90: | ||
|- | |- | ||
|<math>V_{mf}</math> | |<math>V_{mf}</math> | ||
| − | |<math> | + | |<math>3.9 \pm 1.17</math> (n=4)<ref name="Hernandez_2006_model"> Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, ''et al.'' (2006). ''Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase''. FEBS J., 273 , pp. 1975–1988 ([http://dx.doi.org/doi:10.1111/j.1742-4658.2006.05214.x doi]) (Model File) </ref> |
| − | |<math> | + | |<math>mM\cdot(\text{min})^{-1}</math> |
|HeLA cell line | |HeLA cell line | ||
|rowspan="4"| | |rowspan="4"| | ||
Latest revision as of 09:59, 16 October 2014
The enzyme hexokinase phosphorylates (adds a phosphate group to) glucose in the cell's cytoplasm. In the process, a phosphate group from Adenosine triphosphate (ATP) is transferred to glucose producing glucose 6-phosphate (Glc6P).
Contents
Chemical equation
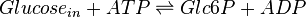
Rate equation
Without inhibition
Random Bi-Bi Michaelis Menten is used.[1]
![v = \frac{\frac{V_{mf}}{Km_{Glucose_{in}}K_{ATP}}\Big( [Glucose_{in}][ATP] - \frac{[Glc6P][ADP]}{K_{eq}} \Big)}{1 + \frac{[Glucose_{in}]}{Km_{Glucose_{in}}} + \frac{[ATP]}{K_{ATP}} + \frac{[Glucose_{in}][ATP]}{Km_{Glucose_{in}}K_{ATP}} + \frac{[Glc6P]}{K_{Glc6P}} + \frac{[ADP]}{K_{ADP}} +\frac{[Glc6P][ADP]}{K_{Glc6P}K_{ADP}} + \frac{[Glucose_{in}][ADP]}{K_{Glucose_{in}}K_{ADP}} +\frac{[Glc6P][ATP]}{K_{Glc6P}K_{ATP}} }](/wiki/images/math/d/2/d/d2d1c3d1115eef8cdf23e7eb98f65b04.png)
With allosteric inhibition
Hexokinase is allosterically inhibited by Glc6P [2]. The rate law taking into account this inhibition is
![\frac{V_{mf}* \frac{[Glucose]*[ATP]}{Km_{Glucose}*Km_{ATP}} }{ \left(1 + \frac{[Glc6P]}{Ki_{Glc6P}} \right)* \left( 1 + \frac{[Glucose]}{Km_{Glucose}} \right) + \frac{[ATP]}{Km_{ATP}} + \frac{[Glucose]*[ATP]}{Km_{Glucose}*Km_{ATP}} }](/wiki/images/math/c/2/6/c26962b0cefb2c26a75d64c2de18328e.png)
Parameters
Without inhibition
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.04 [3] | 
|
HeLa cell line | |

|
651[4] | |||
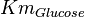
|
0.1[1] | mM | ||
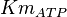
|
1.1[1] | mM | ||
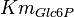
|
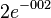 [1] [1]
|
mM | ||
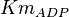
|
3.5[1] | mM |
With inhibition
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
0.04 [3] | 
|
HeLa cell line | |
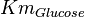
|
0.1[1] | mM | ||
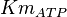
|
1.1[1] | mM | ||
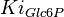
|
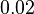 [2] [2]
|
mM |
Parameters with uncertainty
Hexokinase isoenzyme has been reported to vary in different developmental and metabolic status of the cell. In mammalian tissues four isoenzymes of Hexokinase is present [5]. Hexokinase-II has been found to be overexpressed in several first growith cancer cells [3]. Therefore in our model we have considered only the Hexokinase-II kinetic parameter values. The value of Hexokinase is reported as 0.06 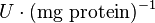 . Converting this value to
. Converting this value to 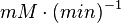 gives the value of 3.9
gives the value of 3.9 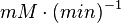 . But in the model file the authors used the value of
. But in the model file the authors used the value of 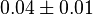 . Same error ratio is used in our model.
. Same error ratio is used in our model.
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
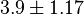 (n=4)[6] (n=4)[6]
|
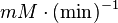
|
HeLA cell line | |
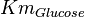
|
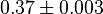 (n=5)[7] (n=5)[7]
|
mM | SNU449 cell (Human Liver Tissue) | |
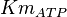
|
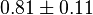 (n=5)[7] (n=5)[7]
|
mM | ||
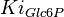
|
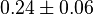 (n=5)[7] (n=5)[7]
|
mM |
Equilibrium constant
| Equilibrium constant | Conditions | Source |
|---|---|---|
| 4908 | pH=7, T=25°C | Voet et al.[8] from Newshole et al. (1973) [9]:
|
| 846 | pH=7, T=25°C | Lehninger, (2008)[10] p 553:
|
| 890 | pH=7, T=25°C | Lehninger, (1975)[11] p 396 |
- Calculating average from these equlibrium constant gives the value of
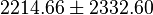
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011) Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 (doi)
- ↑ 2.0 2.1 Wilson J E (2003). Isozymes of mammalian hexokinase: structure, subcellular localization and metabolic function. Journal of Experimental Biology, 206, pp. 2049–2057 (doi)
- ↑ 3.0 3.1 3.2 Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988(doi) Cite error: Invalid
<ref>tag; name "Hernandez_2006" defined multiple times with different content - ↑ Arbitrary value
- ↑ Adams V, Kempf W, Hassam S, Briner J. (1995), Determination of hexokinase isoenzyme I and II by RT-PCR: increased hexokinase II isoenzyme in human renal cell carcinoma. J Biochem Mol Med 1995;54:53–58.
- ↑ Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988 (doi) (Model File)
- ↑ 7.0 7.1 7.2 Ahn, K.J.; Kim, J.; Yun, M.; Park, J.H.; Lee, J.D.(2009), Enzymatic properties of the N- and C-terminal halves of human hexokinase II, BMB Rep. 42, 350-355.
- ↑ Voet, D., Voet., J.G. and Pratt, C. W. (1999) Fundamentals of biochemistry, Wiley
- ↑ Newshole, E.A. and Stuart, C. (1973) Regulation in Metabolism, Wiley
- ↑ David L. Nelson, Michael M. Cox (2008), Lehninger Principles of Biochemistry (5th edn), W. H. Freeman and Company
- ↑ Lehninger, A.L. (1975) Biochemistry (2nd edn), Worth
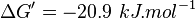 ,
, 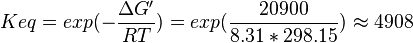
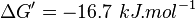 ,
,