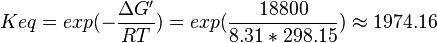Difference between revisions of "3-phosphoglycerate kinase"
(→Parameters with uncertainty) |
(→Parameters with uncertainty) |
||
| (One intermediate revision by the same user not shown) | |||
| Line 66: | Line 66: | ||
|- | |- | ||
|<math>V_{mf}</math> | |<math>V_{mf}</math> | ||
| − | |<math>13 ± 6</math><ref name="Hernandez_2006"></ref> | + | |<math>13 ± 6</math><ref name="Hernandez_2006"></ref> <br> <math>845 ± 390</math><ref name="Hernandez_2006"></ref> |
| − | |<math>mM \times min^{-1}</math> | + | |<math>U\cdot(\text{mg protein})^{-1}</math> <br> <math>mM \times min^{-1}</math> |
|rowspan="6"|Human | |rowspan="6"|Human | ||
|rowspan="6"| | |rowspan="6"| | ||
Latest revision as of 11:01, 16 October 2014
3-Phosphoglycerate kinase (PGK) is an enzyme that catalyzes the reversible transfer of a phosphate group from 1,3-bisphosphoglycerate (1,3-BPG) to ADP producing 3-phosphoglycerate (3-PG) and ATP. Like all kinases it is a transferase.
Contents
Chemical equation
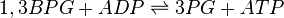
Rate equation
Random Bi-Bi reversible Michaelis-Menten euation for non-interacting substrates are used. [1]
![\frac{V_{mf}\frac{[1,3BPG][ADP]}{K_{1,3BPG} K_{ADP}} - V_{mr}\frac{[3PG][ATP]}{K_{3PG} K_{ATP}}}{1 + \frac{[1,3BPG]}{K_{1,3BPG}} + \frac{[ADP]}{K_{ADP}} + \frac{[1,3BPG][ADP]}{K_{1,3BPG} K_{ADP}} + \frac{[3PG][ATP]}{K_{3PG} K_{ATP}} + \frac{[3PG]}{K_{3PG}} + \frac{[ADP]}{K_{ADP}} }](/wiki/images/math/8/e/b/8eb366ea68004741a7ace7e2cf32d509.png)
Modified rate law to consider thermodynamic constraint
![\frac{V_{mf}\frac{[1,3BPG][ADP]}{K_{1,3BPG} K_{ADP}} \left( 1 - \frac{[3PG][ATP]}{K_{eq} [1,3BPG][ADP]} \right)}{1 + \frac{[1,3BPG]}{K_{1,3BPG}} + \frac{[ADP]}{K_{ADP}} + \frac{[1,3BPG][ADP]}{K_{1,3BPG} K_{ADP}} + \frac{[3PG][ATP]}{K_{3PG} K_{ATP}} + \frac{[3PG]}{K_{3PG}} + \frac{[ADP]}{K_{ADP}} }](/wiki/images/math/0/7/9/079bab3ab4330d6900643599f12e26aa.png)
Parameter values
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
8.7 [2] | 
|
HeLa cell line | |
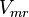
|
2.5[1] | 
| ||
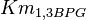
|
0.079[1] | mM | ||
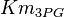
|
0.13[1] | mM | ||
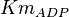
|
0.04[1] | mM | ||
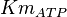
|
0.27[1] | mM |
Parameters with uncertainty
- As the value of the
 does not depend on the organism, the mean and std. dev. of the distribution can be calculated from the various values reported in the literature. [3]
does not depend on the organism, the mean and std. dev. of the distribution can be calculated from the various values reported in the literature. [3]
Alternative: Same percent of error reported for can be considered for
can be considered for 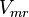 while the mean value as reported in [2] can be considered. In that case the value would be
while the mean value as reported in [2] can be considered. In that case the value would be 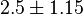
| Parameter | Value | Units | Organism | Remarks |
|---|---|---|---|---|

|
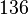 [2] [2] 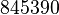 [2] [2]
|
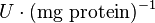 
|
Human | |
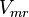
|
Sampled based on Haldane relation or Alternative value | 
| ||
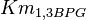
|
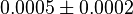 [4] [4]
|
mM | ||
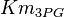 [4] [4]
|
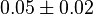
|
mM | ||
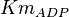
|
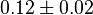 [4] [4]
|
mM | ||
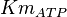
|
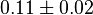 [4] [4]
|
mM |
Equilibrium constant
| Equilibrium constant | Conditions | Source |
|---|---|---|
| 1974.16 | pH=7, T=25°C | Lehninger, (2008)[5] p 553:
|
| 3333 | pH=6.9, T=298.15 K | From Krietsch et al. (1970) [6] |
| 3571 | pH=6.9, T=298.15 K | From Krietsch et al. (1970) [6] |
- Taking average from these values give
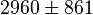
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Marín-Hernández A, Gallardo-Pérez JC, Rodríguez-Enríquez S et al (2011) Modeling cancer glycolysis. Biochim Biophys Acta 1807:755–767 (doi)
- ↑ 2.0 2.1 2.2 2.3 Marín-Hernández A , Rodríguez-Enríquez S, Vital-González P A, et al. (2006). Determining and understanding the control of glycolysis in fast-growth tumor cells. Flux control by an over-expressed but strongly product-inhibited hexokinase. FEBS J., 273 , pp. 1975–1988(doi)
- ↑ Achcar, F., Kerkhoven, E. J., Bakker, B. M., Barrett, M. P., Breitling, R. (2012), Dynamic modelling under uncertainty: the case of Trypanosoma brucei energy metabolism, PLoS Comput. Biol. 8, e1002352.
- ↑ 4.0 4.1 4.2 4.3 Szabo, J.; Varga, A.; Flachner, B.; Konarev, P.V.; Svergun, D.I.; Zavodszky, P.; Vas, M. (2008), Role of side-chains in the operation of the main molecular hinge of 3-phosphoglycerate kinase, FEBS Lett. 582, 1335-1340
- ↑ David L. Nelson, Michael M. Cox (2008), Lehninger Principles of Biochemistry (5th edn), W. H. Freeman and Company
- ↑ 6.0 6.1 Krietsch WZ and Bücher T. (1970) Eur J Biochem. 17(3):568-80. [pmid: 5493986]
 ,
,