Transcription of r
The scbR gene is transcribed into scbR mRNA (r).
Contents
Chemical equation
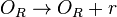
Rate equation
![r= T_{R}\cdot [O_{R}]](/wiki/images/math/9/d/e/9de794e0ab735317530153bc81d570e5.png) (for overlapping promoters)
(for overlapping promoters)![r= \Omega_{R}\cdot [O_{R}]](/wiki/images/math/9/6/c/96ce32f9feedaf5f90408cb4d468db16.png) (for isolated promoters)
(for isolated promoters)Parameters
The parameters of this reaction are the transcription rate of scbR (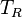 and the firing rate
and the firing rate 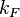 ).
The transcription rate is derived by the strength of the promoter (
).
The transcription rate is derived by the strength of the promoter (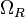 ) but also taking into account the transcriptional interference by the scbA promoter. In this model, we have assumed that the isolated promoter strength is equal to the number of transcripts produced per unit of time. Therefore, the parameter
) but also taking into account the transcriptional interference by the scbA promoter. In this model, we have assumed that the isolated promoter strength is equal to the number of transcripts produced per unit of time. Therefore, the parameter 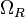 is assumed to be equal to the transcription rate constant of the isolated promoter. These parameter values were derived from published data on E.coli mRNA transcription rate and calculations based on genomic properties of Streptomyces coelicolor A3(2). Additionally, the firing rate (elongation initiation rate) constant (
is assumed to be equal to the transcription rate constant of the isolated promoter. These parameter values were derived from published data on E.coli mRNA transcription rate and calculations based on genomic properties of Streptomyces coelicolor A3(2). Additionally, the firing rate (elongation initiation rate) constant (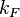 ) is needed to test the strength of each promoter. This parameter is also derived from literature and is sampled from the same distribution for both scbR and scbA promoters, but is then multiplied by a heterogeneity factor
) is needed to test the strength of each promoter. This parameter is also derived from literature and is sampled from the same distribution for both scbR and scbA promoters, but is then multiplied by a heterogeneity factor 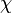 to calculate the final
to calculate the final 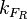 for the scbR promoter. The heterogeneity factor for each promoter is sampled from a log-normal distribution.
for the scbR promoter. The heterogeneity factor for each promoter is sampled from a log-normal distribution.
| Name | Value | Units | Value in previous GBL models [1] [2] | Remarks-Reference |
|---|---|---|---|---|
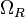
|
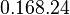 [3] [4] [5] [3] [4] [5]
|

|
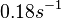
Range tested:
( Bistability range: ( and ( |
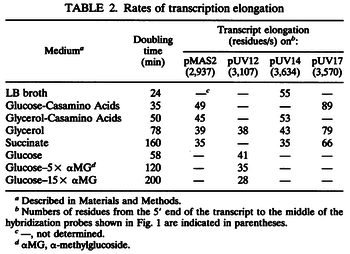
In a recent publication by R.A. Cox, genomic properties and macromolecular compositions of Streptomyces coelicolor A3(2) and E.coli were reported, along with equations that connect these properties. For S. coelicolor, the polypeptide elongation rate 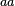 is reported to be in the range between 0.59 and 3.17 amino acids s-1, from which the mRNA elongation rate can be calculated according to Cox from the equation is reported to be in the range between 0.59 and 3.17 amino acids s-1, from which the mRNA elongation rate can be calculated according to Cox from the equation 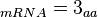 (factor 3 reflects the number of nucleotides per codon), therefore resulting in (factor 3 reflects the number of nucleotides per codon), therefore resulting in 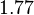 and and 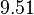 nucleotides nucleotides 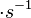 as minimum and maximum values respectively. As ScbR has 648 pb, the transcription rate constant can be calculated as per as minimum and maximum values respectively. As ScbR has 648 pb, the transcription rate constant can be calculated as per 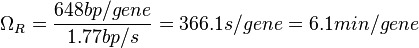 and and 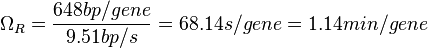 , thus resulting in the final values of , thus resulting in the final values of 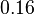 and and 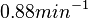 . .
Additionally, Bremer et al. have reported an mRNA transcription rate of 55 noucleotides/s for E. coli, a value which is also shared by R.A. Cox, while Vogel et al. have published a range of mRNA transcription rates in the range of 28-89 noucleotides/s, depending on different growth rates of E. coli. By the same calculations, the corresponding transcription rate constants are |
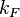
|
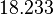 [6] [7] [8] [6] [7] [8]
|

|
N/A | Pai et al. reported a typical transcription initiation rate in QS systems to be 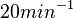 . .
 Pai et al. 2009 [6] This value is also supported by Kennell et al. who calculated the transcription initiation rates from experimental data derived from in vitro experiments using E. coli. The results showed one initiation every 3.3 sec (therefore transcription rate 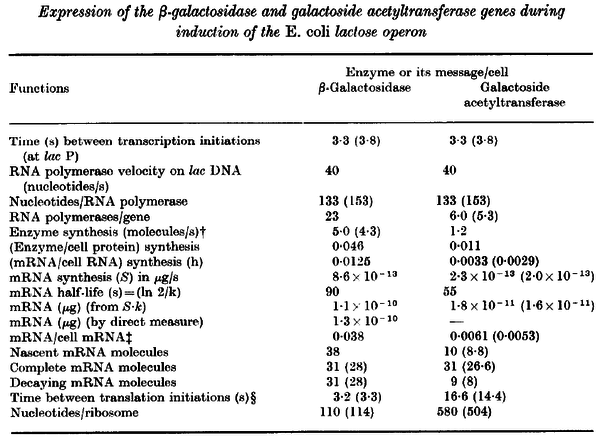 Kennell et al. 1977 [8] Finally, Tadmor et al. reported a maximum transcription initiation rate of 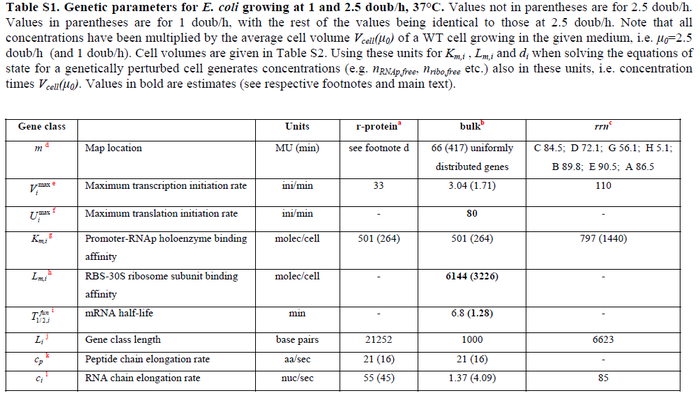 Tadmor et al. 2008 [7] |
In order to include the strength of the promoter in the transcription rate, the following formula is used: 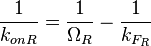 [9]. In this way, a distribution with the values for parameter
[9]. In this way, a distribution with the values for parameter 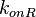 (rate of RNA polymerase binding the promoter) is obtained, which is necessary to link the firing rate with the total transcription rate according to the formulas suggested by Bendtsen et al. [9].
From the
(rate of RNA polymerase binding the promoter) is obtained, which is necessary to link the firing rate with the total transcription rate according to the formulas suggested by Bendtsen et al. [9].
From the 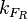 and the
and the 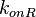 , the maximal occupancy for the isolated scbR promoter can be calculated by using the formula:
, the maximal occupancy for the isolated scbR promoter can be calculated by using the formula: 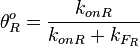 . The promoter aspect ratio
. The promoter aspect ratio 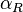 is equal to
is equal to 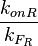 , which leads to the equivalent equation
, which leads to the equivalent equation 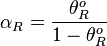 .
The total transcription rate in the case of the isolated (uncoupled) promoters is calculated through the formula:
.
The total transcription rate in the case of the isolated (uncoupled) promoters is calculated through the formula: 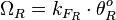 .
Similarly, the maximal occupancy of the isolated scbA promoter (
.
Similarly, the maximal occupancy of the isolated scbA promoter (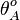 )is calculated as described in the section Transcription of a.
In the case of the overlapping (coupled) promoters the maximal occupancy for the scbR promoter is
)is calculated as described in the section Transcription of a.
In the case of the overlapping (coupled) promoters the maximal occupancy for the scbR promoter is 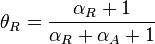 , where
, where 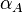 is the promoter aspect ratio for the isolated scbA promoter.
Therefore, the final transcription rate constant is calculated as per
is the promoter aspect ratio for the isolated scbA promoter.
Therefore, the final transcription rate constant is calculated as per 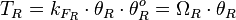 .
.
Parameters with uncertainty
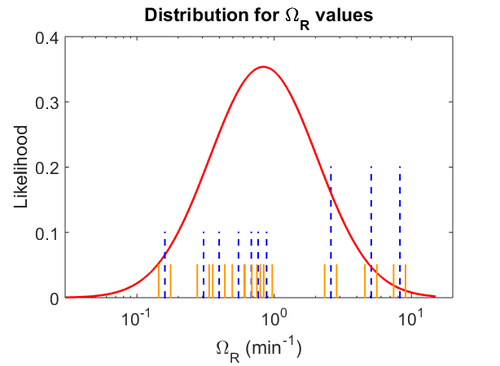
When deciding how to describe the uncertainty for this parameter we must take into consideration that the reported values are either calculated or derived with approximation from experiments and from other macromolecular properties. Additionally, some of the values correspond to mRNA transcription rates of different bacteria species (E. coli). This means that there might be a notable difference between actual parameter values and the ones reported in literature. These facts influence the quantification of the parameter uncertainty and therefore the shape of the corresponding distributions. Therefore, by assigning the appropriate weights to the parameter values and using the method described here, the appropriate probability distributions were designed.
Although the weight of the distribution is put on the values calculated for S. coelicolor by setting 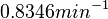 as the mode of the log-normal distribution for the
as the mode of the log-normal distribution for the 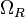 , we wish to explore the full range of reported values. Thus, the Spread is set to
, we wish to explore the full range of reported values. Thus, the Spread is set to 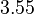 and the range where 68.27% of the values are found is between
and the range where 68.27% of the values are found is between 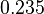 and
and 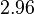
 .
.
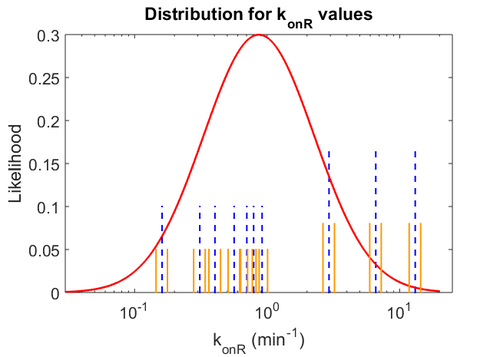
With regards to the firing rate 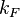 , the reported values are within the range of
, the reported values are within the range of 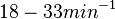 with the most probable values being
with the most probable values being 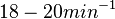 . Since these values are reported as being the average rates (and
. Since these values are reported as being the average rates (and 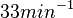 being the maximum), we will also sample lower values, so the final sampling range will be around the values
being the maximum), we will also sample lower values, so the final sampling range will be around the values 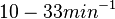 . The mode of the distribution is set to
. The mode of the distribution is set to 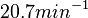 and the Spread is set to
and the Spread is set to 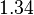 . Therefore, the range where 68.27% of the values are found is between
. Therefore, the range where 68.27% of the values are found is between 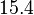 and
and 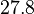
 .
.
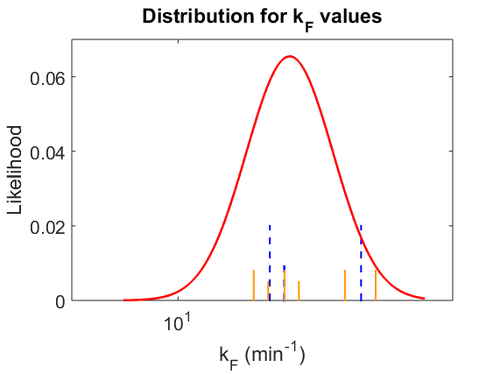
From the literature values of 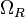 and
and 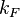 , the values for
, the values for 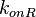 are calculated. By assigning the same weights to each of the
are calculated. By assigning the same weights to each of the 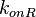 values as of the
values as of the 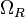 values, a distribution with a mode of
values, a distribution with a mode of 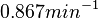 and a Spread of
and a Spread of 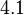 is generated. The range where 68.27% of the values are found is between
is generated. The range where 68.27% of the values are found is between 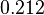 and
and 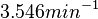 .
.
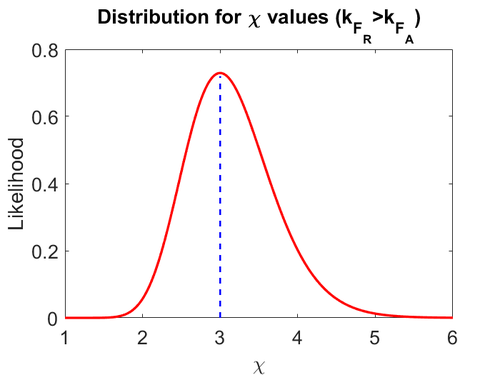
Finally, the heterogeneity factor can be set so that 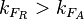 or
or 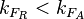 . In the first case,
. In the first case, 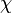 has values within the range 1-10 and in the second within the range 0.1-0.9. In order for the two promoters to have equal strength,
has values within the range 1-10 and in the second within the range 0.1-0.9. In order for the two promoters to have equal strength, 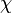 is set to be equal to 1.
Therefore, the mode of the heterogeneity factor
is set to be equal to 1.
Therefore, the mode of the heterogeneity factor 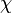 is
is 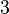 and the Spread is
and the Spread is 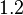 (68.27% of the values are found between
(68.27% of the values are found between 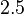 and
and 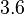 ), in the case where scbR promoter is stronger. In the opposite case the mode is
), in the case where scbR promoter is stronger. In the opposite case the mode is 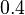 and the Spread is
and the Spread is 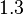 (68.27% of the values are found between
(68.27% of the values are found between 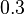 and
and 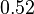 ).
).
The probability distributions for the parameters, adjusted accordingly in order to reflect the above values, are the following:
The values retrieved from literature and their weights are indicated by the blue dashed lines, and the uncertainty for each value is indicated using the reported experimental error (green lines) or a default value of 10% error (orange lines).
The parameter information of the distributions is:
| Parameter | Mode | Spread | μ | σ |
|---|---|---|---|---|
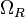
|
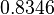
|
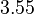
|
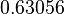
|
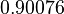
|
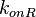
|
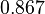
|
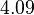
|
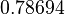
|
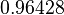
|
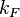
|
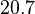
|
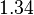
|
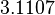
|
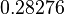
|
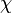 ( (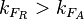 ) )
|
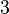
|
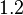
|
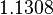
|
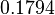
|
References
- ↑ 1.0 1.1 1.2 S. Mehra, S. Charaniya, E. Takano, and W.-S. Hu. A bistable gene switch for antibiotic biosynthesis: The butyrolactone regulon in streptomyces coelicolor. PLoS ONE, 3(7), 2008.
- ↑ 2.0 2.1 2.2 A. Chatterjee, L. Drews, S. Mehra, E. Takano, Y.N. Kaznessis, and W.-S. Hu. Convergent transcription in the butyrolactone regulon in streptomyces coelicolor confers a bistable genetic switch for antibiotic biosynthesis. PLoS ONE, 6(7), 2011.
- ↑ 3.0 3.1 3.2 Cox RA. Quantitative relationships for specific growth rates and macromolecular compositions of Mycobacterium tuberculosis, Streptomyces coelicolor A3(2) and Escherichia coli B/r: an integrative theoretical approach. Microbiology. 2004 May;150(Pt 5):1413-26.
- ↑ 4.0 4.1 Bremer H., Yuan D. RNA chain growth-rate in Escherichia coli, Journal of Molecular Biology, 1968; 38:(2), p. 163-180
- ↑ 5.0 5.1 Vogel U., Jensen KF. The RNA chain elongation rate in Escherichia coli depends on the growth rate. Journal of Bacteriology. 1994;176(10):2807-2813.
- ↑ 6.0 6.1 Pai, A. and You, L. Optimal tuning of bacterial sensing potential. Mol Syst Biol. 2009; 5: 286
- ↑ 7.0 7.1 Tadmor AD, Tlusty T. A Coarse-Grained Biophysical Model of E. coli and Its Application to Perturbation of the rRNA Operon Copy Number. PLoS Comput Biol (2008); 4(5): e1000038. doi: 10.1371/journal.pcbi.1000038
- ↑ 8.0 8.1 Kennell D., Riezman H. Transcription and translation initiation frequencies of the Escherichia coli lac operon. J. Mol. Biol. 1977; 114(1):1-21
- ↑ 9.0 9.1 Bendtsen KM, Erdőssy J, Csiszovszki Z, et al. Direct and indirect effects in the regulation of overlapping promoters. Nucleic Acids Research. 2011;39(16):6879-6885. doi:10.1093/nar/gkr390.



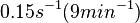
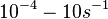
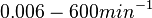 )
)
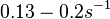
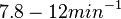 )
)
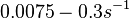
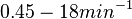 )
)
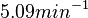 and
and 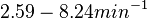 .
.
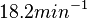 ).
).